Assurance de qualité pour le cancer rectal – phase 2 ...
Assurance de qualité pour le cancer rectal – phase 2 ...
Assurance de qualité pour le cancer rectal – phase 2 ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
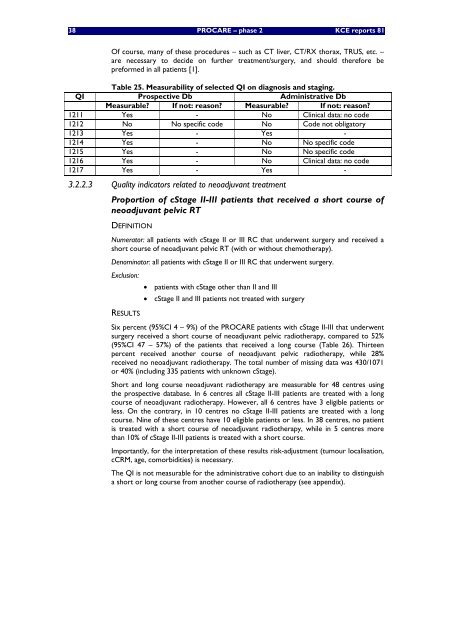
38 PROCARE <strong>–</strong> <strong>phase</strong> 2 KCE reports 81Of course, many of these procedures <strong>–</strong> such as CT liver, CT/RX thorax, TRUS, etc. <strong>–</strong>are necessary to <strong>de</strong>ci<strong>de</strong> on further treatment/surgery, and should therefore bepreformed in all patients [1].Tab<strong>le</strong> 25. Measurability of se<strong>le</strong>cted QI on diagnosis and staging.QI Prospective Db Administrative DbMeasurab<strong>le</strong>? If not: reason? Measurab<strong>le</strong>? If not: reason?1211 Yes - No Clinical data: no co<strong>de</strong>1212 No No specific co<strong>de</strong> No Co<strong>de</strong> not obligatory1213 Yes - Yes -1214 Yes - No No specific co<strong>de</strong>1215 Yes - No No specific co<strong>de</strong>1216 Yes - No Clinical data: no co<strong>de</strong>1217 Yes - Yes -3.2.2.3 Quality indicators related to neoadjuvant treatmentProportion of cStage II-III patients that received a short course ofneoadjuvant pelvic RTDEFINITIONNumerator: all patients with cStage II or III RC that un<strong>de</strong>rwent surgery and received ashort course of neoadjuvant pelvic RT (with or without chemotherapy).Denominator: all patients with cStage II or III RC that un<strong>de</strong>rwent surgery.Exclusion:• patients with cStage other than II and III• cStage II and III patients not treated with surgeryRESULTSSix percent (95%CI 4 <strong>–</strong> 9%) of the PROCARE patients with cStage II-III that un<strong>de</strong>rwentsurgery received a short course of neoadjuvant pelvic radiotherapy, compared to 52%(95%CI 47 <strong>–</strong> 57%) of the patients that received a long course (Tab<strong>le</strong> 26). Thirteenpercent received another course of neoadjuvant pelvic radiotherapy, whi<strong>le</strong> 28%received no neoadjuvant radiotherapy. The total number of missing data was 430/1071or 40% (including 335 patients with unknown cStage).Short and long course neoadjuvant radiotherapy are measurab<strong>le</strong> for 48 centres usingthe prospective database. In 6 centres all cStage II-III patients are treated with a longcourse of neoadjuvant radiotherapy. However, all 6 centres have 3 eligib<strong>le</strong> patients or<strong>le</strong>ss. On the contrary, in 10 centres no cStage II-III patients are treated with a longcourse. Nine of these centres have 10 eligib<strong>le</strong> patients or <strong>le</strong>ss. In 38 centres, no patientis treated with a short course of neoadjuvant radiotherapy, whi<strong>le</strong> in 5 centres morethan 10% of cStage II-III patients is treated with a short course.Importantly, for the interpretation of these results risk-adjustment (tumour localisation,cCRM, age, comorbidities) is necessary.The QI is not measurab<strong>le</strong> for the administrative cohort due to an inability to distinguisha short or long course from another course of radiotherapy (see appendix).