Conference, Proceedings
Conference, Proceedings
Conference, Proceedings
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
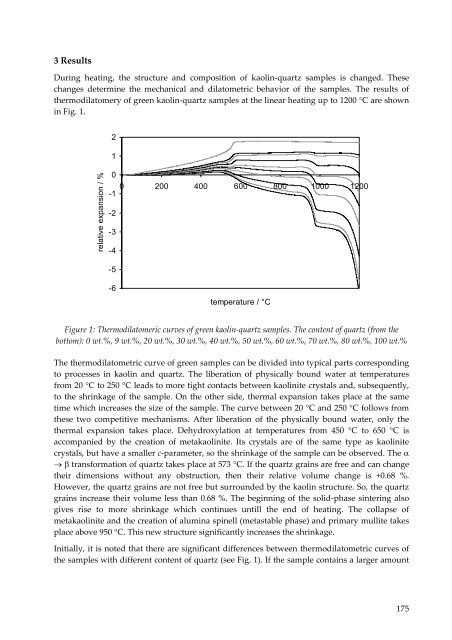
3 Results<br />
During heating, the structure and composition of kaolin‐quartz samples is changed. These<br />
changes determine the mechanical and dilatometric behavior of the samples. The results of<br />
thermodilatomery of green kaolin‐quartz samples at the linear heating up to 1200 °C are shown<br />
in Fig. 1.<br />
relative expansion / %<br />
2<br />
1<br />
0<br />
-1<br />
-2<br />
-3<br />
-4<br />
-5<br />
-6<br />
0 200 400 600 800 1000 1200<br />
temperature / °C<br />
Figure 1: Thermodilatomeric curves of green kaolin‐quartz samples. The content of quartz (from the<br />
bottom): 0 wt.%, 9 wt.%, 20 wt.%, 30 wt.%, 40 wt.%, 50 wt.%, 60 wt.%, 70 wt.%, 80 wt.%, 100 wt.%<br />
The thermodilatometric curve of green samples can be divided into typical parts corresponding<br />
to processes in kaolin and quartz. The liberation of physically bound water at temperatures<br />
from 20 °C to 250 °C leads to more tight contacts between kaolinite crystals and, subsequently,<br />
to the shrinkage of the sample. On the other side, thermal expansion takes place at the same<br />
time which increases the size of the sample. The curve between 20 °C and 250 °C follows from<br />
these two competitive mechanisms. After liberation of the physically bound water, only the<br />
thermal expansion takes place. Dehydroxylation at temperatures from 450 °C to 650 °C is<br />
accompanied by the creation of metakaolinite. Its crystals are of the same type as kaolinite<br />
crystals, but have a smaller c‐parameter, so the shrinkage of the sample can be observed. The α<br />
→ β transformation of quartz takes place at 573 °C. If the quartz grains are free and can change<br />
their dimensions without any obstruction, then their relative volume change is +0.68 %.<br />
However, the quartz grains are not free but surrounded by the kaolin structure. So, the quartz<br />
grains increase their volume less than 0.68 %. The beginning of the solid‐phase sintering also<br />
gives rise to more shrinkage which continues untill the end of heating. The collapse of<br />
metakaolinite and the creation of alumina spinell (metastable phase) and primary mullite takes<br />
place above 950 °C. This new structure significantly increases the shrinkage.<br />
Initially, it is noted that there are significant differences between thermodilatometric curves of<br />
the samples with different content of quartz (see Fig. 1). If the sample contains a larger amount<br />
175