Research Report 2010 2011 - Helmholtz-Zentrum für ...
Research Report 2010 2011 - Helmholtz-Zentrum für ...
Research Report 2010 2011 - Helmholtz-Zentrum für ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
RESEARCH REVIEWS | The Aging of the Immune System: Challenges and Perspectives<br />
31<br />
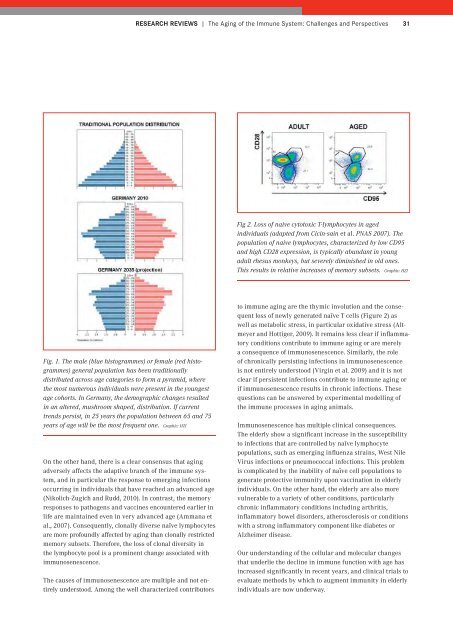
Fig 2. Loss of naive cytotoxic T-lymphocytes in aged<br />
individuals (adapted from Cicin-sain et al. PNAS 2007). The<br />
population of naive lymphocytes, characterized by low CD95<br />
and high CD28 expression, is typically abundant in young<br />
adult rhesus monkeys, but severely diminished in old ones.<br />
This results in relative increases of memory subsets. Graphic: HZI<br />
Fig. 1. The male (blue histogrammes) or female (red histogrammes)<br />
general population has been traditionally<br />
distributed across age categories to form a pyramid, where<br />
the most numerous individuals were present in the youngest<br />
age cohorts. In Germany, the demographic changes resulted<br />
in an altered, mushroom shaped, distribution. If current<br />
trends persist, in 25 years the population between 65 and 75<br />
years of age will be the most frequent one. Graphic: HZI<br />
On the other hand, there is a clear consensus that aging<br />
adversely affects the adaptive branch of the immune system,<br />
and in particular the response to emerging infections<br />
occurring in individuals that have reached an advanced age<br />
(Nikolich-Zugich and Rudd, <strong>2010</strong>). In contrast, the memory<br />
responses to pathogens and vaccines encountered earlier in<br />
life are maintained even in very advanced age (Ammana et<br />
al., 2007). Consequently, clonally diverse naïve lymphocytes<br />
are more profoundly affected by aging than clonally restricted<br />
memory subsets. Therefore, the loss of clonal diversity in<br />
the lymphocyte pool is a prominent change associated with<br />
immunosenescence.<br />
The causes of immunosenescence are multiple and not entirely<br />
understood. Among the well characterized contributors<br />
to immune aging are the thymic involution and the consequent<br />
loss of newly generated naïve T cells (Figure 2) as<br />
well as metabolic stress, in particular oxidative stress (Altmeyer<br />
and Hottiger, 2009). It remains less clear if inflammatory<br />
conditions contribute to immune aging or are merely<br />
a consequence of immunosenescence. Similarly, the role<br />
of chronically persisting infections in immunosenescence<br />
is not entirely understood (Virgin et al. 2009) and it is not<br />
clear if persistent infections contribute to immune aging or<br />
if immunosenescence results in chronic infections. These<br />
questions can be answered by experimental modelling of<br />
the immune processes in aging animals.<br />
Immunosenescence has multiple clinical consequences.<br />
The elderly show a significant increase in the susceptibility<br />
to infections that are controlled by naïve lymphocyte<br />
populations, such as emerging influenza strains, West Nile<br />
Virus infections or pneumococcal infections. This problem<br />
is complicated by the inability of naïve cell populations to<br />
generate protective immunity upon vaccination in elderly<br />
individuals. On the other hand, the elderly are also more<br />
vulnerable to a variety of other conditions, particularly<br />
chronic inflammatory conditions including arthritis,<br />
inflammatory bowel disorders, atherosclerosis or conditions<br />
with a strong inflammatory component like diabetes or<br />
Alzheimer disease.<br />
Our understanding of the cellular and molecular changes<br />
that underlie the decline in immune function with age has<br />
increased significantly in recent years, and clinical trials to<br />
evaluate methods by which to augment immunity in elderly<br />
individuals are now underway.