Nucleotide Analogs - Jena Bioscience
Nucleotide Analogs - Jena Bioscience
Nucleotide Analogs - Jena Bioscience
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
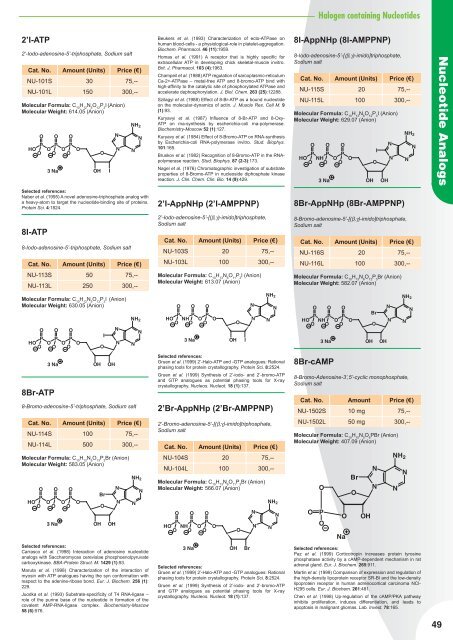
2’I-ATP<br />
2’-Iodo-adenosine-5’-triphosphate, Sodium salt<br />
Cat. No. Amount (Units) Price (€)<br />
NU-101S 30 75,--<br />
NU-101L 150 300,--<br />
Molecular Formula: C 10 H 12 N 5 O 12 P 3 I (Anion)<br />
Molecular Weight: 614.05 (Anion)<br />
O<br />
O O<br />
P P P<br />
HO O O O<br />
O O O<br />
3 Na<br />
O<br />
OH I<br />
N<br />
N<br />
NH 2<br />
Selected references:<br />
Naber et al. (1995) A novel adenosine-triphosphate analog with<br />
a heavy-atom to target the nucleotide-binding site of proteins.<br />
Protein Sci. 4:1824.<br />
8I-ATP<br />
8-Iodo-adenosine-5’-triphosphate, Sodium salt<br />
Cat. No. Amount (Units) Price (€)<br />
NU-113S 50 75,--<br />
NU-113L 250 300,--<br />
Molecular Formula: C 10 H 12 N 5 O 13 P 3 I (Anion)<br />
Molecular Weight: 630.05 (Anion)<br />
O<br />
3 Na<br />
O O<br />
P P P<br />
HO O O O<br />
O O O<br />
8Br-ATP<br />
I<br />
O<br />
OH OH<br />
8-Bromo-adenosine-5’-triphosphate, Sodium salt<br />
N<br />
N<br />
N<br />
N<br />
N<br />
NH2<br />
Cat. No. Amount (Units) Price (€)<br />
NU-114S 100 75,--<br />
NU-114L 500 300,--<br />
Molecular Formula: C 10 H 12 N 5 O 13 P 3 Br (Anion)<br />
Molecular Weight: 583.05 (Anion)<br />
O<br />
3 Na<br />
O O<br />
P P P<br />
HO O O O<br />
O O O<br />
Br<br />
O<br />
OH OH<br />
N<br />
N<br />
N<br />
N<br />
NH2<br />
Selected references:<br />
Carrasco et al. (1998) Interaction of adenosine nucleotide<br />
analogs with Saccharomyces cerevisiae phosphoenolpyruvate<br />
carboxykinase. BBA-Protein Struct. M. 1429 (1):93.<br />
Maruta et al. (1998) Characterization of the interaction of<br />
myosin with ATP analogues having the syn conformation with<br />
respect to the adenine-ribose bond. Eur. J. Biochem. 256 (1):<br />
229.<br />
Juodka et al. (1993) Substrate-specifi city of T4 RNA-ligase –<br />
role of the purine base of the nucleotide in formation of the<br />
covalent AMP-RNA-ligase complex. Biochemistry-Moscow<br />
58 (6):576.<br />
N<br />
Beukers et al. (1993) Characterization of ecto-ATPase on<br />
human blood-cells - a physiological-role in platelet-aggregation.<br />
Biochem. Pharmacol. 46 (11):1959.<br />
Homas et al. (1991) A receptor that is highly specifi c for<br />
extracellular ATP in developing chick skeletal-muscle invitro.<br />
Brit. J. Pharmacol. 103 (4):1963.<br />
Champeil et al. (1988) ATP regulation of sarcoplasmic-reticulum<br />
Ca-2+-ATPase – metal-free ATP and 8-bromo-ATP bind with<br />
high-affi nity to the catalytic site of phosphorylated ATPase and<br />
accelerate dephosphorylation. J. Biol. Chem. 263 (25):12288.<br />
Szilagyi et al. (1988) Effect of 8-Br-ATP as a bound nucleotide<br />
on the molecular-dynamics of actin. J. Muscle Res. Cell M. 9<br />
(1):93.<br />
Kuryavyi et al. (1987) Infl uence of 8-Br-ATP and 8-Oxy-<br />
ATP on rna-synthesis by escherichia-coli rna-polymerase.<br />
Biochemistry-Moscow 52 (1):127.<br />
Kuryavy et al. (1984) Effect of 8-Bromo-ATP on RNA-synthesis<br />
by Escherichia-coli RNA-polymerase invitro. Stud. Biophys.<br />
101:165.<br />
Bruskov et al. (1982) Recognition of 8-Bromo-ATP in the RNApolymerase<br />
reaction. Stud. Biophys. 87 (2-3):173.<br />
Nagel et al. (1976) Chromatographic investigation of substrate<br />
properties of 8-Bromo-ATP in nucleoside diphosphate kinase<br />
reaction. J. Clin. Chem. Clin. Bio. 14 (9):429.<br />
2’I-AppNHp (2’I-AMPPNP)<br />
2’-Iodo-adenosine-5’-[(β,γ)-imido]triphosphate,<br />
Sodium salt<br />
Cat. No. Amount (Units) Price (€)<br />
NU-103S 20 75,--<br />
NU-103L 100 300,--<br />
Molecular Formula: C 10 H 13 N 6 O 11 P 3 I (Anion)<br />
Molecular Weight: 613.07 (Anion)<br />
O<br />
O O<br />
P P P<br />
HO NH O<br />
O O O<br />
3 Na<br />
O<br />
O<br />
OH I<br />
N<br />
N<br />
NH 2<br />
Selected references:<br />
Gruen et al. (1999) 2’-Halo-ATP and -GTP analogues: Rational<br />
phasing tools for protein crystallography. Protein Sci. 8:2524.<br />
Gruen et al. (1999) Synthesis of 2’-iodo- and 2’-bromo-ATP<br />
and GTP analogues as potential phasing tools for X-ray<br />
crystallography. Nucleos. Nucleot. 18 (1):137.<br />
2’Br-AppNHp (2’Br-AMPPNP)<br />
2’-Bromo-adenosine-5’-[(β,γ)-imido]triphosphate,<br />
Sodium salt<br />
Cat. No. Amount (Units) Price (€)<br />
NU-104S 20 75,--<br />
NU-104L 100 300,--<br />
Molecular Formula: C 10 H 13 N 6 O 11 P 3 Br (Anion)<br />
Molecular Weight: 566.07 (Anion)<br />
O<br />
O O<br />
P P P<br />
HO NH O<br />
O O O<br />
3 Na<br />
O<br />
O<br />
OH Br<br />
N<br />
N<br />
N<br />
N<br />
N<br />
NH 2<br />
Selected references:<br />
Gruen et al. (1999) 2’-Halo-ATP and -GTP analogues: Rational<br />
phasing tools for protein crystallography. Protein Sci. 8:2524.<br />
Gruen et al. (1999) Synthesis of 2’-iodo- and 2’-bromo-ATP<br />
and GTP analogues as potential phasing tools for X-ray<br />
crystallography. Nucleos. Nucleot. 18 (1):137.<br />
N<br />
8I-AppNHp (8I-AMPPNP)<br />
8-Iodo-adenosine-5’-[(β,γ)-imido]triphosphate,<br />
Sodium salt<br />
Cat. No. Amount (Units) Price (€)<br />
NU-115S 20 75,--<br />
NU-115L 100 300,--<br />
Molecular Formula: C 10 H 13 N 6 O 12 P 3 I (Anion)<br />
Molecular Weight: 629.07 (Anion)<br />
O<br />
O O<br />
P P P<br />
HO NH O<br />
O O O<br />
3 Na<br />
O<br />
I<br />
O<br />
OH OH<br />
8Br-AppNHp (8Br-AMPPNP)<br />
8-Bromo-adenosine-5’-[(β,γ)-imido]triphosphate,<br />
Sodium salt<br />
N<br />
N<br />
NH 2<br />
Cat. No. Amount (Units) Price (€)<br />
NU-116S 20 75,--<br />
NU-116L 100 300,--<br />
Molecular Formula: C 10 H 13 N 6 O 12 P 3 Br (Anion)<br />
Molecular Weight: 582.07 (Anion)<br />
O<br />
O O<br />
P P P<br />
HO NH O<br />
O O O<br />
3 Na<br />
8Br-cAMP<br />
O<br />
Br<br />
O<br />
OH OH<br />
N<br />
N<br />
N<br />
N<br />
NH 2<br />
8-Bromo-Adenosine-3’,5’-cyclic monophosphate,<br />
Sodium salt<br />
Cat. No. Amount Price (€)<br />
NU-1502S 10 mg 75,--<br />
NU-1502L 50 mg 300,--<br />
Molecular Formula: C 10 H 10 N 5 O 6 PBr (Anion)<br />
Molecular Weight: 407.09 (Anion)<br />
O<br />
Halogen containing <strong>Nucleotide</strong>s<br />
O<br />
P<br />
O<br />
O<br />
Na<br />
Br<br />
O<br />
OH<br />
N<br />
N<br />
NH2<br />
Selected references:<br />
Paz et al. (1999) Corticotropin increases protein tyrosine<br />
phosphatase activity by a cAMP-dependent mechanism in rat<br />
adrenal gland. Eur. J. Biochem. 265:911.<br />
Martin et al. (1999) Comparison of expression and regulation of<br />
the high-density lipoprotein receptor SR-BI and the low-density<br />
lipoprotein receptor in human adrenocortical carcinoma NCI-<br />
H295 cells. Eur. J. Biochem. 261:481.<br />
Chen et al. (1998) Up-regulation of the cAMP/PKA pathway<br />
inhibits proliferation, induces differentiation, and leads to<br />
apoptosis in malignant gliomas. Lab. Invest. 78:165.<br />
N<br />
N<br />
N<br />
N<br />
49<br />
<strong>Nucleotide</strong> <strong>Analogs</strong>