Raport de cercetare - Lorentz JÄNTSCHI
Raport de cercetare - Lorentz JÄNTSCHI
Raport de cercetare - Lorentz JÄNTSCHI
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
0.9974 0.9948 2<br />
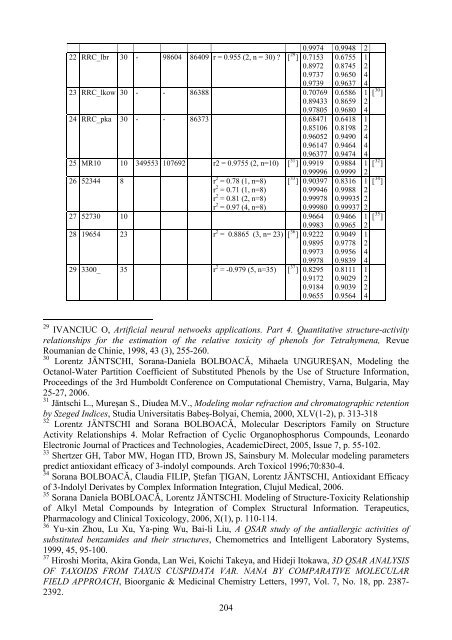
22 RRC_lbr 30 - 98604<br />
29<br />
86409 r = 0.955 (2, n = 30) ? [ ] 0.7153 0.6755 1<br />
0.8972 0.8745 2<br />
0.9737 0.9650 4<br />
0.9739 0.9637 4<br />
23 RRC_lkow 30 - - 86388 0.70769 0.6586<br />
30<br />
1 [ ]<br />
0.89433 0.8659 2<br />
0.97805 0.9680 4<br />
24 RRC_pka 30 - - 86373 0.68471 0.6418 1<br />
0.85106 0.8198 2<br />
0.96052 0.9490 4<br />
0.96147 0.9464 4<br />
0.96377 0.9474 4<br />
25 MR10 10 349553 107692 r2 = 0.9755 (2, n=10)<br />
31<br />
[ ] 0.9919 0.9884<br />
32<br />
1 [ ]<br />
0.99996 0.9999 2<br />
26 52344 8 r 2 = 0.78 (1, n=8)<br />
33<br />
[ ] 0.90397 0.8316<br />
34<br />
1 [ ]<br />
r 2 = 0.71 (1, n=8) 0.99946 0.9988 2<br />
r 2 = 0.81 (2, n=8) 0.99978 0.99935 2<br />
r 2 = 0.97 (4, n=8) 0.99980 0.99937 2<br />
27 52730 10 0.9664 0.9466<br />
35<br />
1 [ ]<br />
0.9983 0.9965 2<br />
28 19654 23 r 2 = 0.8865 (3, n= 23)<br />
36<br />
[ ] 0.9222 0.9049 1<br />
0.9895 0.9778 2<br />
0.9973 0.9956 4<br />
0.9978 0.9839 4<br />
29 3300_ 35 r 2 = -0.979 (5, n=35)<br />
37<br />
[ ] 0.8295 0.8111 1<br />
0.9172 0.9029 2<br />
0.9184 0.9039 2<br />
0.9655 0.9564 4<br />
29 IVANCIUC O, Artificial neural netwoeks applications. Part 4. Quantitative structure-activity<br />
relationships for the estimation of the relative toxicity of phenols for Tetrahymena, Revue<br />
Roumanian <strong>de</strong> Chinie, 1998, 43 (3), 255-260.<br />
30 <strong>Lorentz</strong> <strong>JÄNTSCHI</strong>, Sorana-Daniela BOLBOACĂ, Mihaela UNGUREŞAN, Mo<strong>de</strong>ling the<br />
Octanol-Water Partition Coefficient of Substituted Phenols by the Use of Structure Information,<br />
Proceedings of the 3rd Humboldt Conference on Computational Chemistry, Varna, Bulgaria, May<br />
25-27, 2006.<br />
31 Jäntschi L., Mureşan S., Diu<strong>de</strong>a M.V., Mo<strong>de</strong>ling molar refraction and chromatographic retention<br />
by Szeged Indices, Studia Universitatis Babeş-Bolyai, Chemia, 2000, XLV(1-2), p. 313-318<br />
32 <strong>Lorentz</strong> <strong>JÄNTSCHI</strong> and Sorana BOLBOACĂ, Molecular Descriptors Family on Structure<br />
Activity Relationships 4. Molar Refraction of Cyclic Organophosphorus Compounds, Leonardo<br />
Electronic Journal of Practices and Technologies, Aca<strong>de</strong>micDirect, 2005, Issue 7, p. 55-102.<br />
33 Shertzer GH, Tabor MW, Hogan ITD, Brown JS, Sainsbury M. Molecular mo<strong>de</strong>ling parameters<br />
predict antioxidant efficacy of 3-indolyl compounds. Arch Toxicol 1996;70:830-4.<br />
34 Sorana BOLBOACĂ, Claudia FILIP, Ştefan ŢIGAN, <strong>Lorentz</strong> <strong>JÄNTSCHI</strong>, Antioxidant Efficacy<br />
of 3-Indolyl Derivates by Complex Information Integration, Clujul Medical, 2006.<br />
35 Sorana Daniela BOBLOACĂ, <strong>Lorentz</strong> <strong>JÄNTSCHI</strong>. Mo<strong>de</strong>ling of Structure-Toxicity Relationship<br />
of Alkyl Metal Compounds by Integration of Complex Structural Information. Terapeutics,<br />
Pharmacology and Clinical Toxicology, 2006, X(1), p. 110-114.<br />
36 Yu-xin Zhou, Lu Xu, Ya-ping Wu, Bai-li Liu, A QSAR study of the antiallergic activities of<br />
substituted benzami<strong>de</strong>s and their structures, Chemometrics and Intelligent Laboratory Systems,<br />
1999, 45, 95-100.<br />
37 Hiroshi Morita, Akira Gonda, Lan Wei, Koichi Takeya, and Hi<strong>de</strong>ji Itokawa, 3D QSAR ANALYSIS<br />
OF TAXOIDS FROM TAXUS CUSPIDATA VAR. NANA BY COMPARATIVE MOLECULAR<br />
FIELD APPROACH, Bioorganic & Medicinal Chemistry Letters, 1997, Vol. 7, No. 18, pp. 2387-<br />
2392.<br />
204