Thesis Title: Subtitle - NMR Spectroscopy Research Group
Thesis Title: Subtitle - NMR Spectroscopy Research Group
Thesis Title: Subtitle - NMR Spectroscopy Research Group
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
68 Chapter 2. Possum: paramagnetically orchestrated spectral solver of unassigned methyls.<br />
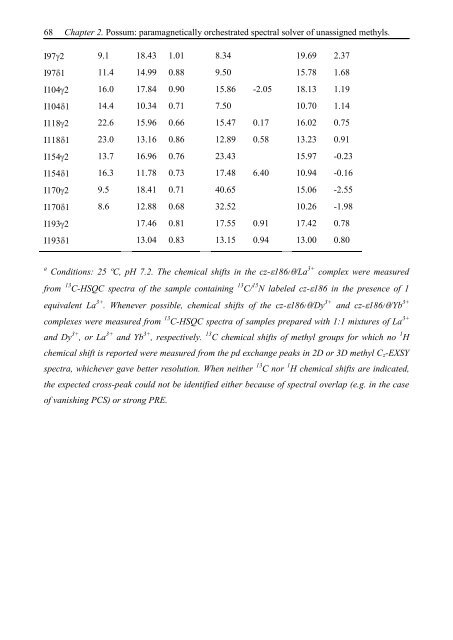
I97 2 9.1 18.43 1.01 8.34 19.69 2.37<br />
I97 1 11.4 14.99 0.88 9.50 15.78 1.68<br />
I104 2 16.0 17.84 0.90 15.86 -2.05 18.13 1.19<br />
I104 1 14.4 10.34 0.71 7.50 10.70 1.14<br />
I118 2 22.6 15.96 0.66 15.47 0.17 16.02 0.75<br />
I118 1 23.0 13.16 0.86 12.89 0.58 13.23 0.91<br />
I154 2 13.7 16.96 0.76 23.43 15.97 -0.23<br />
I154 1 16.3 11.78 0.73 17.48 6.40 10.94 -0.16<br />
I170 2 9.5 18.41 0.71 40.65 15.06 -2.55<br />
I170 1 8.6 12.88 0.68 32.52 10.26 -1.98<br />
I193 2 17.46 0.81 17.55 0.91 17.42 0.78<br />
I193 1 13.04 0.83 13.15 0.94 13.00 0.80<br />
a Conditions: 25 ºC, pH 7.2. The chemical shifts in the cz- 186/ /La 3+ complex were measured<br />
from 13 C-HSQC spectra of the sample containing 13 C/ 15 N labeled cz- 186 in the presence of 1<br />
equivalent La 3+ . Whenever possible, chemical shifts of the cz- 186/ /Dy 3+ and cz- 186/ /Yb 3+<br />
complexes were measured from 13 C-HSQC spectra of samples prepared with 1:1 mixtures of La 3+<br />
and Dy 3+ , or La 3+ and Yb 3+ , respectively. 13 C chemical shifts of methyl groups for which no 1 H<br />
chemical shift is reported were measured from the pd exchange peaks in 2D or 3D methyl Cz-EXSY<br />
spectra, whichever gave better resolution. When neither 13 C nor 1 H chemical shifts are indicated,<br />
the expected cross-peak could not be identified either because of spectral overlap (e.g. in the case<br />
of vanishing PCS) or strong PRE.