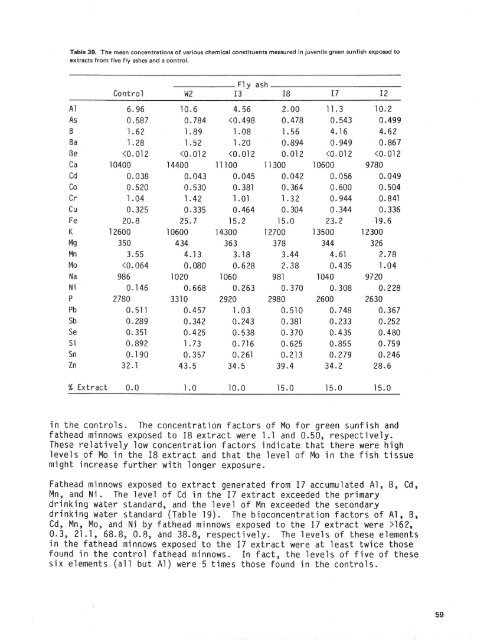
The mean concentrations <strong>of</strong> various chemical constituents measured in juvenile green sunfish exposed toextracts from five <strong>fly</strong> <strong>ash</strong>es <strong>and</strong> a control.Fly <strong>ash</strong>Control W2 I3 I8 I7 I2% Extract 0-0in the controls- The concentration factors <strong>of</strong> o for green sunfish <strong>and</strong>fathead minnows exposed to I8 extract were 1.1 nd 0.50, respectively.These relatively low concentration factors indicate that there were higho in the I8 extract <strong>and</strong> that the level <strong>of</strong> o in the fish tissuemight increase further with longer exposure.Fathead minnow osed to extract generated from I7 accumulated Al, B, Cd,in the I7 ext act exceeded the primarythe level <strong>of</strong> n exceeded the secondaryj The bioconcentration factors <strong>of</strong> Al, B,nows exposed to the I7 extract were >162,.8, respectively, The levels <strong>of</strong> these elementsin the fathead minnows exposed to the I7 extract were at least twice thosefound in the control fathead minnows. In fact, the levels <strong>of</strong> five <strong>of</strong> thesesix elements (all but Al) were 5 times tin the controls.
The green sunfish exposed to t t accumulated e same elementsas the fathead minnows althoug ese constitue accumulated to alesser degree.resent in the green sunfishexposed to thecontrols. Theexposed to the IFinally, the composition <strong>of</strong> the extracts generated from samples I7 <strong>and</strong> I2were similar. The accumulation <strong>of</strong> elements Prom the I2 extract by the testorganisms also was similar to that observe in test organisms exposed to17. The chemical constituents accumul ated to the reatest degree by thefathead minnows were B, Cd, Mn, NO, <strong>and</strong> 8). The concentrationfactors for 5, Cd, Mn, Ma, <strong>and</strong> Ni in fat posed to the 12extract were 0.3, >Q.3, 81.8, L2, <strong>and</strong> >22.1, respectively.The green sunfish exposed to the I2 extract accumulated the same chemicalcsnsti tuents awere accumul atsunf i sh expose7.0 times, respectively,those measuredors for B <strong>and</strong> Mo ingreen sunfishThe six most frequently accumulated chemical constituents from the <strong>fly</strong> <strong>ash</strong>extracts were Al, B, C , Mn, MO, <strong>and</strong> i, Other chemical constituents wereaccumulated, but these elements were accumulated to the greatest extent.In most situations, the results <strong>of</strong> the chemical analyses <strong>of</strong> the extracts<strong>and</strong> the LC-50 determinations did not indicate which chemical constituentswould be accumulated. The United States Food <strong>and</strong> Drug Admini tration (FDA)currently lists Hg, Pb, Cd, As, Se, <strong>and</strong> Zn at the top <strong>of</strong> the riority listin its program concerning toxic elements in food (Jelinek <strong>and</strong> Cornel i ussen,77). Of these, only Hg has an FDB-specified regulatory limit for fishd shellfish (Anonymous, 1974); FDA guidelines for other metals in foodshave no% been establishe (Phi l l ips <strong>and</strong> Russo, 1978).Aluminum is an element<strong>and</strong> it rarely prAluminum has a ronsumption <strong>of</strong>the low toxicity <strong>of</strong> A1is relatively inert on biological processes,raeder <strong>and</strong> Darndency in fresBoron is used in a process for bleaching ulverized wood by the pulp <strong>and</strong>paper industry (Thompson et al., l976), a(Phi 11 ips <strong>and</strong> Russo, l978), <strong>and</strong> as a neutroinstallations (National Academy <strong>of</strong> Science,<strong>fly</strong> <strong>ash</strong> from fossil fuels. Boron generallytendency in freshwater fish <strong>and</strong> a low toxicity to aquatic organisms <strong>and</strong> tohurnans (Phi 11 ips <strong>and</strong> Russo, 1978).Cadmium is rare in nature, but is highly toxic (National Academy <strong>of</strong>Science, 1973). Inhalation or ingestion <strong>of</strong> Cd produces both acute <strong>and</strong>chronic health effects. Cadmium poisonings in urnans resulting from oralconsumption or inhalation are well documented ( assett, 1975; Flick et al.,1971; Voors <strong>and</strong> Shuman, 1977; American ConIndustrial Hygientists, 1974; Stokinger, 1
- Page 1 and 2:
lllinoiSTATSTAT
- Page 3 and 4:
V SURVEYatural Resources Building60
- Page 5 and 6:
TABLESSummary of the origin and gen
- Page 7 and 8:
The overall purpose of this investi
- Page 9 and 10:
steady state conditions in the disp
- Page 11 and 12:
total carbon determinations were ca
- Page 13 and 14: y passing the extract through a col
- Page 15 and 16: The GC-MS analyses were performed b
- Page 17 and 18: with ultrapure water. The final HCl
- Page 19 and 20: Be 3. Wlir~eraiogical composi'tion
- Page 21 and 22: Table 6. Trace constituent concentr
- Page 23 and 24: Table 7. Fly ash sample classificat
- Page 25 and 26: Table 8. Carbon, sulfur, and benzen
- Page 27 and 28: Wave number (cm-'Figure 8. Infrared
- Page 29 and 30: Figure 12. HPLC chromatogram of the
- Page 31 and 32: Figure 16. HPLC chromatogram of the
- Page 34 and 35: Table 11. Organic components in the
- Page 36 and 37: Figure 21. Gas chromatogram of the
- Page 38 and 39: 4.Jrcrm c o mQU"* 'tn U.a-U U O NrO
- Page 40 and 41: constituents. The amount of soluble
- Page 42 and 43: Table 14. Change in chemical compos
- Page 44 and 45: Table 16. Change in chemical compos
- Page 46 and 47: Change in chemical composition as a
- Page 48 and 49: Figure 26. Changes in the concentra
- Page 50 and 51: Table 19. Corytituents in the long-
- Page 52 and 53: Table 21. The LC-50 values, amount
- Page 54 and 55: Table 23. The range of concentratio
- Page 56 and 57: Table 25. The range of concentratio
- Page 58 and 59: Table 27. The mean initial lengths
- Page 60 and 61: Table 32. The mean final lengths an
- Page 62 and 63: Several of the extrbivalves ( ~ are
- Page 66 and 67: 1972). Cadmium is a dangerous cumul
- Page 68 and 69: Bl umer, M., 1957, Removal of eleme
- Page 70 and 71: Furr, A. K-, T. F, Parkinson, R. A.
- Page 72 and 73: Murtha, M. J., and G. Burnet, 1979,
- Page 74 and 75: Standard methods for examination of