SOP â Malaria Microscopy - NVBDCP
SOP â Malaria Microscopy - NVBDCP
SOP â Malaria Microscopy - NVBDCP
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
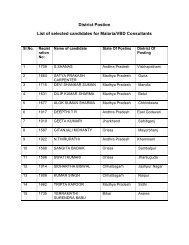
Quality Assurance of <strong>Malaria</strong> Diagnostic testsAt each level, based on the results received in the proforma given at Form 10 followingactions have to be taken8.7.4.3.1 Regional Reference Laboratories (ROH & FW) :-• compare results of malaria microscopy of PHC laboratories with the known key.• analyse results and identifiy the quality problems.• analyse problems and provide feedback to respective laboratory.• send quarterly reports to NIMR.8.7.4.3.2 National Reference Laboratory (NIMR):-• compare results of EQAS panel of slides received from RRLs.• analyse EQAS results of RRLs and the PHC level, received quarterly from the ROH& FW and identify the quality problems.• analysis of problems and feed back.• quarterly report to Dte. of <strong>NVBDCP</strong>.8.7.4.3.3 In collaboration with <strong>NVBDCP</strong> the NRL would undertake the following• develop solution to the problems for improving quality.• evaluate the quality improvements after EQAS implementation.• dispatch the feed back and suggest solutions to PHC laboratory through ROH & FWsand respective SPOs/DMOs.8.7.5 The EQAS results would be referred to for accreditation of the LTs.8.7.6 ConfidentialityDo not, under any circumstances, divulge the results of the tests tounauthorized individuals and results of one laboratory to another.<strong>SOP</strong> – <strong>Malaria</strong> <strong>Microscopy</strong>© Copyright to Dte. <strong>NVBDCP</strong> Only. Any modification are prohibited