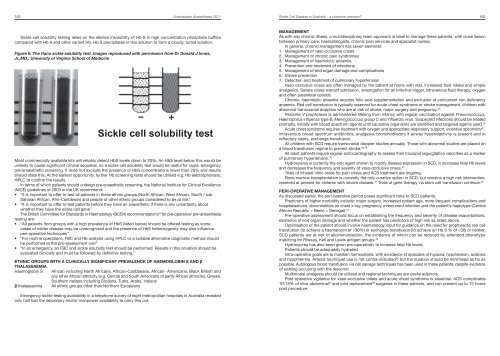
152 <strong>Australasian</strong> <strong>Anaesthesia</strong> <strong>2011</strong>Sickle Cell Disease in Australia – a phantom menace? 153Sickle cell solubility testing relies on the relative insolubility of Hb S in high concentration phosphate bufferscompared with Hb A <strong>and</strong> other variant Hb. Hb S precipitates in this solution to form a cloudy, turbid solution.Figure 6. The Itano sickle solubility test. Images reproduced with permission from Dr Donald J.Innes,Jr.,MD,: University of Virginia School of MedicineSickle cell solubility testMost commercially available kits will reliably detect HbS levels down to 20%. An HbS level below this would beunlikely to cause significant clinical sequelae, so a sickle cell solubility test would be useful for rapid, emergencypre-anaesthetic screening. It does not exclude the presence of HbS concentrations lower than 20% <strong>and</strong> resultsshould state this. At the earliest opportunity, further Hb screening tests should be utilised e.g. Hb electrophoresis,HPLC to confirm the results.In terms of which patients should undergo pre-anaesthetic screening, the National Institute for Clinical Excellence(NICE) guidelines of 2003 in the UK recommend:• “It is important to offer to test all patients in these ethnic groups (North African, West African, South / subSaharan African, Afro-Caribbean) <strong>and</strong> people of other ethnic groups considered to be at risk”• “It is important to offer to test patients before they have an anaesthetic if there is any uncertainty aboutwhether they have the sickle cell gene”The British Committee for St<strong>and</strong>ards in Haematology (BCSH) recommendations 24 for pre-operative/ pre-anaesthesiatesting are:• “All patients from groups with a high prevalence of HbS (listed below) should be offered testing as somecases of milder disease may be unrecognized <strong>and</strong> the presence of HbS heterozygosity may also influenceperi-operative techniques”• “For routine operations, FBC <strong>and</strong> Hb analysis using HPLC or a suitable alternative diagnostic method shouldbe performed at the pre-assessment visit”• “In an emergency, an FBC <strong>and</strong> sickle solubility test should be performed. Results in this situation should beevaluated clinically <strong>and</strong> must be followed by definitive testing.”ETHNIC GROUPS WITH A CLINICALLY SIGNIFICANT PREVALENCE OF HAEMOGLOBIN Ѕ AND βTHALASSAEMIAHaemoglobin S African including North Africans, African-Caribbeans, African- Americans, Black British <strong>and</strong>any other African ethnicity (e.g. Central <strong>and</strong> South Americans of partly African ethnicity), Greeks,Southern Italians including Sicilians, Turks, Arabs, Indiansβ thalassaemia All ethnic groups other than Northern EuropeansEmergency sickle testing availability in a telephone survey of eight metropolitan hospitals in Australia revealedonly half had the laboratory <strong>and</strong>/or manpower availability to carry this out.MANAGEMENTAs with any chronic illness, a multidisciplinary team approach is ideal to manage these patients, with close liaisonbetween primary care, haematologists, chronic pain services <strong>and</strong> specialist nurses.In general, chronic management has seven elements:1. Management of vaso-occlusive crises2. Management of chronic pain syndromes3. Management of haemolytic anaemia4. Prevention <strong>and</strong> treatment of infections5. Management of end organ damage <strong>and</strong> complications6. Stroke prevention7. Detection <strong>and</strong> treatment of pulmonary hypertensionVaso-occlusive crises are often managed by the patient at home with rest, increased fluid intake <strong>and</strong> simpleanalgesics. Severe crises warrant admission, investigation for an infective trigger, intravenous fluid therapy, oxygen<strong>and</strong> often parenteral opioids.Chronic, haemolytic anaemia requires folic acid supplementation <strong>and</strong> exclusion of concurrent iron deficiencyanaemia. Red cell transfusion is typically reserved for acute chest syndrome or stroke management, children withabnormal transcranial dopplers who are at risk of stroke, major surgery <strong>and</strong> pregnancy. 25Penicillin V prophylaxis is administered lifelong from infancy with regular vaccination against Pneumococcus,Haemophilus influenza type B, Meningococcus group C <strong>and</strong> influenza virus. Suspected infections should be treatedpromptly, initially with broad spectrum agents until causative organisms are identified <strong>and</strong> targeted agents used. 26Acute chest syndrome requires treatment with oxygen <strong>and</strong> appropriate respiratory support, incentive spirometry 27 ,intravenous broad spectrum antibiotics, analgesia, bronchodilators if airway hyperreactivity is present <strong>and</strong> inrefractory cases, exchange transfusion.All children with SCD require transcranial doppler studies annually. Those with abnormal studies are placed on28 29a blood transfusion regime to prevent stroke.All adult patients require regular echocardiography to assess their tricuspid regurgitation velocities as a markerof pulmonary hypertension. 30Hydroxyurea is currently the only agent shown to modify disease expression in SCD. It increases fetal Hb levels<strong>and</strong> decreases the frequency <strong>and</strong> severity of vaso-occlusive crises. 31Trials of inhaled nitric oxide for pain crises <strong>and</strong> ACS treatment are ongoing.Bone marrow transplantation is currently the only curative option in SCD, but remains a high risk intervention,reserved at present for children with severe disease. 32 Trials of gene therapy via stem cell transfusion continue. 33PERI-OPERATIVE MANAGEMENTAs discussed earlier, the peri-operative period poses significant risks to SCD patients.Predictors of higher morbidity include: major surgery, increased patient age, more frequent complications <strong>and</strong>hospitalisations, abnormalities on chest x-ray, pregnancy, intercurrent infection <strong>and</strong> the patient’s haplotype (CentralAfrican Republic > Benin > Senegal). 34Pre-operative assessment should focus on establishing the frequency <strong>and</strong> severity of disease exacerbations,existence of end organ damage <strong>and</strong> whether the patient has predictors of high risk as listed above.Optimisation of the patient should involve haematology input for guidance on the need for prophylactic red celltransfusion (to achieve a haematocrit of >30%) or exchange transfusion (to achieve an Hb S % of
154 <strong>Australasian</strong> <strong>Anaesthesia</strong> <strong>2011</strong>Sickle Cell Disease in Australia – a phantom menace? 155CONCLUSIONSDecisions regarding which individuals should be tested for sickle cell disease should be informed by consensusguidelines based on disease patterns in the general populace. In the US, since the late 1980’s, universal neonatalscreening has been advocated by the Agency for Health Care Policy (AHCPR). The agency stated that targetingonly high risk racial groups would not identify all affected infants, as health officials could not reliably determine aninfant’s race by appearance, name or parental report.Analysing disease risk by presumed ethnic origin alone presents a potentially dangerous oversimplification.Migration of black Africans to Britain has been documented since Roman times, resulting in genetic mixing that isnot obviously apparent.A group of “indigenous British” men from Yorkshire were found to have genetic markers originating from WestAfrica. 39In the UK over 10 years ago, a case of unexpected sickle cell trait emerged in a white woman with no discernableAfrican heritage. On donating blood, she was notified by the National Blood Service about her sickle cell trait status.Further investigation revealed she was descended from a Jamaican slave who had lived in Liverpool in the 18 thcentury. The story made the UK national press <strong>and</strong> was not an isolated case.With growing evidence of this genetic disease in a population not previously thought to be at risk, it is now UKpolicy to universally screen all newborns for sickle cell disease, regardless of their presumed ethnic origin. This isaside from the increased numbers of interracial relationships, bringing together heterozygote alleles from differentpopulations, be it sickle or the thalassaemias.DOES THIS EXPERIENCE EXTRAPOLATE TO THE AUSTRALIAN POPULATION?Certainly, historically at-risk groups are present in the <strong>Australian</strong> population <strong>and</strong> their numbers will only increasewith further immigration. In addition, ethnic groups with other clinically significant haemoglobinopathies, such asthe α <strong>and</strong> β thalassaemias, are well established in the community, <strong>and</strong> it is not inconceivable that with populationmixing, presentations of double heterozygotes will increase.Knowledge within the wider medical community needs to increase in line with this change. These patients willpresent to primary care <strong>and</strong> to other specialities of secondary care aside from haematology. They require lifelongsurveillance, prophylaxis, vaccination <strong>and</strong> psychosocial support to manage their chronic illness as well asknowledgeable clinicians to manage acute complications <strong>and</strong> adequately prepared peri-operative teams to safelynegotiate surgery <strong>and</strong> anaesthesia.REFERENCES1. Ashley-Koch A, Yang Q, Olney RS. Sickle Hemoglobin (HbS) Allel <strong>and</strong> Sickle Cell Disease: A HuGE Review.American Journal of Epidemiology 2000; 151 (9): 839-845.2. Nagel RL, Ranney HM. Genetic epidemiology of structural mutations of the beta-globin gene. Semin Hematol1990: 27:342-59.3. Thomas PW, Higgs DR, Serjeant GR. Benign clinical course in homozygous sickle cell disease: a search forpredictors. J Clin Epidemiol 1997; 50:121-6.4. Dresbach M. Elliptical human red corpuscles. Science 1904; 19: 469-70.5. Dresbach M. Elliptical human red corpuscles (a supplementary statement). Science 1905; 21: 473-5.6. Mason VR. Sickle cell anemia. J Am Med Assoc 1922; 79:1318-20.7. Pauling L, Itano HA, Singer SJ, Wells IC. Sickle cell anemia. Science 1949; 110: 543-8.8. Pagnier J, Mears JG, Dunda-Belkodja O, Schaefer-Rego KE, Beldjord C, Nagel RL, Labie D. Evidence for themulticentric origin of the sickle cell hemoglobin gene in Africa. PNAS. 1984;81 (6):1771-73.9. Aluoch JR. Higher resistance to Plasmodium falciparum infection in patients with homozygous sickle cell diseasein western Kenya. Trap Med Int Health 1997;2:568-71.10. World Health Organisation. Sickle-cell anaemia – Report by the Secretariat to the 59 th World Health Assembly2006.11. Government of Western Australia, Department of local government <strong>and</strong> regional development(http://www.dlgrd.wa.gov.au/Publications/Docs/StatSnapshot_populationDemographics.asp).12. Harley JD, Concannon AJ. Eleven cases of sickle cell disease in Sydney. Med J Aust 1978; 2(9): 401-4.13. Firth PG. <strong>Anaesthesia</strong> for peculiar cells-a century of sickle cell disease. British Journal of <strong>Anaesthesia</strong>.2005;95(3):287-99.14. Hebbel RP, Vercelloti GM. The endothelial biology of sickle cell disease. J Lab Clin Med 1997; 129: 288-93.15. Lancaster JR jr. Reaping of nitric oxide by sickle cell disease. Proc Natl Acad Sci USA 2002; 99:552-3.16. Vichinsky EP, Neumayr LD, Earles AN, et al. Causes <strong>and</strong> outcomes of the acute chest syndrome in sickle celldisease. National Acute Chest Syndrome Study Group. N Eng J Med 2000; 342:1855-65.17. Ataga K, Sood N, De Gent G, et al. Pulmonary hypertension in sickle cell disease. Am J Med 2004;117(9):665-9.18. Eckman JR. Leg ulcers in sickle cell disease. Hematol Oncol Clin North Am 1996;10(6):1333-44.19. Quinn CT et al. Prognostic significance of early vaso-occlusive complications in children with sickle cell anaemia.Blood 2007:109(1):40-45.20. Smith JA. Bone disorders in sickle cell disease. Hematol Oncol Clin North Am 1996;10(6):1345-56.21. Saborio P, Scheinman JI. Sickle cell nephropathy. J Am Soc Nephrol 1999;10(1):187-92.22. Koshy M, Weiner SJ, Miller ST, et al. Surgery <strong>and</strong> anaesthesia in sickle cell disease. Cooperative Study of SickleCell Diseases. Blood 1995; 86: 3676-84.23. Bain BJ. 2006. Haemoglobinopathy Diagnosis. 2 nd ed. London: Blackwell Publishing.24. Ryan K et al. Significant haemoglobinopathies: guidelines for screening <strong>and</strong> diagnosis. British Committee forSt<strong>and</strong>ards in Haematology writing group 2009. British Journal of Haematology 2010; 149: 35-49.25. Reed WF, Vichinsky EP. Transfusion practice for patients with sickle cell disease. Curr Opin Hematol1999;6(6):432-6.26. Overturf GD. Infections <strong>and</strong> immunizations of children with sickle cell disease. Adv Pediatr Infect Dis. 1999;14:191-218.27. Bellet PS et al. Incentive spirometry to prevent acute pulmonary complications in sickle cell diseases.N Engl J Med 1995;333:699-703.28. Adams RJ, McKie VC, Hsu L, et al. Prevention of a first stroke by transfusions in children with sickle cell anemia<strong>and</strong> abnormal results on transcranial Doppler ultrasonography. N Engl J Med 1998;339(1):5-11.29. Adams RJ. Lessons from the Stroke Prevention Trial in Sickle Cell Anaemia (STOP) study. J Child Neurol 2000;15(5):344-9.30. Gladwin MT, Vichinsky EP. Pulmonary complications of sickle cell disease. N Engl J Med 2008;359:2254-2265.31. Platt OS. Hydroxyurea for the treatment of sickle cell anaemia. N Engl J Med 2008;358:1362-1369.32. Vermylen C, Cornu G. Bone marrow transplantation for sickle cell anemia. Curr Opin Hematol. Mar 1996;3(2):163-6.33. Miniero R, Rocha V, Saracco P, et al. Cord blood transplantation (CBT) in hemoglobinopathies. Eurocord.Bone Marrow Transplant 1998;22 Suppl 1:S78-9.34. Firth PG, Head A. Sickle cell disease <strong>and</strong> <strong>Anaesthesia</strong>. Anaesthesiology 2004;101:766-85.35. Moreira Junior G, Bordin JO, Kuroda A, Kerbauy J. Red blood cell alloimmunization in sickle cell disease:theinfluence of racial <strong>and</strong> antigenic pattern differences between donors <strong>and</strong> recipients in Brazil. Am J Hematol1996;52:197-200.36. Stein RE, Urbaniak J. Use of the tourniquet during surgery in patients with sickle cell hemoglobinopathies.Clin Orthop 1980;151:231-3.37. Haberkern CM, Neumayr LD, Orringer EP et al. Cholecystectomy in sickle cell anemia patients: perioperativeoutcome of 364 cases from the National Preoperative Transfusion Study. Preoperative Transfusion in Sickle CellDisease Study Group. Blood 1997;89:1533-42.38. Vichinsky EP, Neumayr LD, Haberkern C, et al. The perioperative complication rate of orthopaedic surgery insickle cell disease:report of the National Sickle Cell Surgery Study Group. Am J Haematol 1999;62:129-38.39. King TE, Parkin EJ et al. Africans in Yorkshire? The deepest-rooting clade of the Y phylogeny within an Englishgeneaology. Eur J Hum Genet 2007;15(3):288-93.