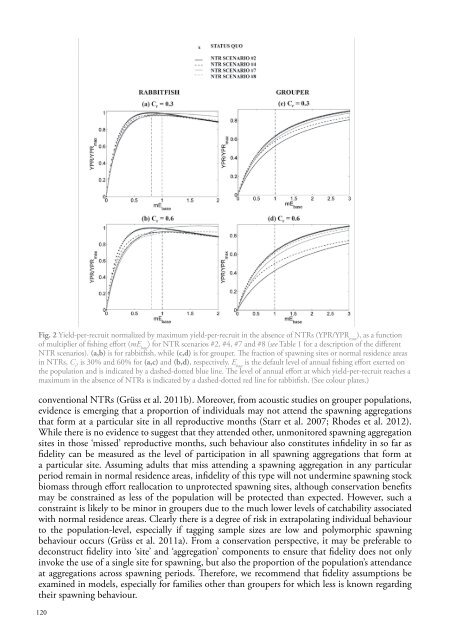
Fig. 2 Yield-per-recruit normalized by maximum yield-per-recruit in the absence of NTRs (YPR/YPR max), as a functionof multiplier of fishing effort (mE base) for NTR scenarios #2, #4, #7 and #8 (see Table 1 for a description of the differentNTR scenarios). (a,b) is for rabbitfish, while (c,d) is for grouper. The fraction of <strong>spawning</strong> sites or normal residence areasin NTRs, C r, is 30% and 60% for (a,c) and (b,d), respectively. E baseis the default level of annual fishing effort exerted onthe population and is indicated by a dashed-dotted blue line. The level of annual effort at which yield-per-recruit reaches amaximum in the absence of NTRs is indicated by a dashed-dotted red line for rabbitfish. (See colour plates.)conventional NTRs (Grüss et al. 2011b). Moreover, from acoustic studies on grouper populations,evidence is emerging that a proportion of individuals may not attend the <strong>spawning</strong> aggregationsthat form at a particular site in all reproductive months (Starr et al. 2007; Rhodes et al. 2012).While there is no evidence to suggest that they attended other, unmonitored <strong>spawning</strong> aggregationsites in those ‘missed’ reproductive months, such behaviour also constitutes infidelity in so far asfidelity can be measured as the level of participation in all <strong>spawning</strong> aggregations that form ata particular site. Assuming adults that miss attending a <strong>spawning</strong> aggregation in any particularperiod remain in normal residence areas, infidelity of this type will not undermine <strong>spawning</strong> stockbiomass through effort reallocation to unprotected <strong>spawning</strong> sites, although conservation benefitsmay be constrained as less of the population will be protected than expected. However, such aconstraint is likely to be minor in groupers due to the much lower levels of catchability associatedwith normal residence areas. Clearly there is a degree of risk in extrapolating individual behaviourto the population-level, especially if tagging sample sizes are low and polymorphic <strong>spawning</strong>behaviour occurs (Grüss et al. 2011a). From a conservation perspective, it may be preferable todeconstruct fidelity into ‘site’ and ‘aggregation’ components to ensure that fidelity does not onlyinvoke the use of a single site for <strong>spawning</strong>, but also the proportion of the population’s attendanceat aggregations across <strong>spawning</strong> periods. Therefore, we recommend that fidelity assumptions beexamined in models, especially for families other than groupers for which less is known regardingtheir <strong>spawning</strong> behaviour.120
When the effort that was in reserves before they were closed disappears or is displaced to non<strong>spawning</strong>sites, setting aside <strong>spawning</strong> sites as reserves reduces sex ratio bias for protogynouspopulations and therefore may increase the chances of egg fertilization (Coleman et al. 1996;Rhodes and Warren-Rhodes 2005). However, if only a small fraction of <strong>spawning</strong> aggregation sitesis protected, fish are faithful to <strong>spawning</strong> sites and effort is displaced to non-protected <strong>spawning</strong>sites, the sex ratio of a part of the population is normalized, while that of the rest of the populationbecomes severely biased towards females. The finding that only partial protection of <strong>spawning</strong>sites can in fact worsen sex ratio has important practical implications in that fisher knowledge andtargeted fishing often extends to aggregations at locations unknown to scientists and managers(Pears et al. 2007; Robinson et al. 2011). Effort displacement rather than disappearance is commonupon creation of reserves (Valcic 2009) and, in the case of aggregation-based NTRs, effort can bereadily displaced to unprotected <strong>spawning</strong> sites (Rhodes and Warren-Rhodes 2005) or equallyvulnerable migratory routes (Claro and Lindeman 2003; Rhodes and Tupper 2008; Rhodes et al.2012). Hence, seasonal prohibitions of take, possession or sale of groupers are expected to be moreeffective for conservation in cases where few <strong>spawning</strong> sites are known to managers.The relative difference in catchability between <strong>spawning</strong> and non-<strong>spawning</strong> periods is a criticaldeterminant of population vulnerability to targeted aggregation fishing (Robinson et al. 2011).In some species, non-<strong>spawning</strong> populations are of extremely low density, or are inaccessible, andcatches outside the <strong>spawning</strong> season are rare. The formation of <strong>spawning</strong> aggregations greatlyincreases density and may result in large changes to catchability if aggregation sites are accessible.This is the case for the Nassau (Epinephelus striatus) and tiger (Mycteroperca tigris) groupers in thetropical western Atlantic (Sadovy and Eklund 1999; Matos-Caraballo et al. 2006). It also appearsthat Epinephelus lanceolatus in Unguja, Zanzibar, is subject to similar dramatic population andfishery changes, whereby catchability increases by orders of magnitude at <strong>spawning</strong> sites comparedto negligible levels in non-<strong>spawning</strong> periods. For most other aggregative spawners, including ourstudy populations of E. fuscoguttatus, E. polyphekadion and S. sutor, catchability does not differ tothe same extent and annual catches comprise fish taken at <strong>spawning</strong> sites and fish taken from thenormal areas of residence; i.e. there is less relative change in catchability between <strong>spawning</strong> andnon-<strong>spawning</strong> periods. Catchability underlies the finding that female SSBR and sex ratio wererelatively unchanged between the different fidelity scenarios. The creation of NTRs eliminates aportion of fishing mortality, but our study populations were still exposed to considerable fishingmortality outside of the <strong>spawning</strong> season, thereby reducing the negative impact of infidelity relativeto populations that are only caught while aggregating to spawn (Grüss et al. in press).As expected, the positive effects of <strong>spawning</strong> aggregation-based NTRs on fish reproductive capacityand their negative effects on yield-per-recruit are stronger for long-lived, slow-growing populationsthan for short-lived, fast-growing populations. This finding offers avenues for prioritisingconservation and management objectives among species. In the case of protogynous groupers,which appear to show relatively high if not absolute fidelity, a precautionary approach would entailprotecting <strong>spawning</strong> aggregations through spatial or non-spatial measures and aiming for moderateYPR targets. Since a significant proportion of <strong>spawning</strong> sites would require protection to achieveconservation benefits (i.e. maintain SSBR), non-spatial measures, such as seasonal sales bans, arelikely to be more effective.In the case of the more productive rabbitfish populations, maximum YPR can be targeted as amanagement objective. Since <strong>spawning</strong> aggregation-based NTRs will neither achieve this objectivenor the conservation objective of maintaining stock biomass, especially given a degree of infidelity,control of fishing effort is the most viable management measure. Given that effort is difficultto regulate in small-scale multi-species and multi-gear fisheries (McClanahan et al. 2005a), analternative approach would be to reduce juvenile mortality through gear measures that increaseselectivity for larger fish or establishment of NTRs in juvenile habitats, though in the longer term,annual effort may increase in unprotected areas including <strong>spawning</strong> aggregations, potentiallyresulting in growth overfishing.121
- Page 4:
The designation of geographical ent
- Page 9:
Chapter 1: IntroductionJan Robinson
- Page 12 and 13:
limited, subsistence levels of expl
- Page 14:
NTRs for spawning aggregations usin
- Page 17 and 18:
al. 2003). Verification may include
- Page 19 and 20:
a fraction of spawning sites are pr
- Page 21 and 22:
Chapter 3: Targeted fishing of the
- Page 23 and 24:
verifying spawning aggregations, we
- Page 25 and 26:
ecorded from inshore close to the c
- Page 27 and 28:
(a)(b)Fig. 3. Spatial patterns ofca
- Page 30 and 31:
pooled sizes of the three spawning
- Page 32 and 33:
found S. sutor contributed up to 44
- Page 34 and 35:
2011b). However, observations of fi
- Page 36 and 37:
MethodsTo identify seasonal and lun
- Page 38 and 39:
n=199Females GSI (mean ± SE)2.521.
- Page 40 and 41:
The estimate of size at maturity in
- Page 42 and 43:
This study was designed to verify S
- Page 44 and 45:
were selected. Fish selected for ta
- Page 46 and 47:
The number of traps increased on th
- Page 48 and 49:
Of the 9 tagged fish detected by re
- Page 50 and 51:
Fig. 7. Diel patterns ofdetection f
- Page 52 and 53:
Spawning aggregation site fidelity
- Page 54 and 55:
Chapter 6: Shoemaker spinefoot rabb
- Page 56 and 57:
anterior of the anus and below the
- Page 58 and 59:
A high percentage (80.8%) of depart
- Page 60 and 61:
arrivals and departures at these tw
- Page 62 and 63:
are typically applied for reef fish
- Page 64 and 65:
(a)(b)(c)Chapter 3, Figure 3. Spati
- Page 66 and 67:
(1)(2)(3)(4)(5)(6)Chapter 7, Table
- Page 68 and 69:
Chapter 12, Fig. 1 Fraction of fema
- Page 70 and 71:
Plates 8. Selected photographs from
- Page 72 and 73:
MethodsStudy sitesThe study area wa
- Page 74 and 75:
which shelved gently ( ca. 25 o ) t
- Page 76 and 77:
Fig. 4. Lunar periodicity in number
- Page 78 and 79: Behaviour and appearanceDescription
- Page 80 and 81: eported aggregations forming betwee
- Page 82 and 83: The sizes of E. fuscoguttatus aggre
- Page 84 and 85: Materials and methodsStudy area and
- Page 86 and 87: TL. All fish tagged were considered
- Page 88 and 89: Lunar timing of arrivals and depart
- Page 90 and 91: Fig. 8. The presence and absence of
- Page 92 and 93: aggregation fishing. This critical
- Page 94 and 95: Chapter 9: Persistence of grouper (
- Page 96 and 97: ResultsBetween 2003 and 2006, the c
- Page 98 and 99: Fig. 2. Mean (± standard error, SE
- Page 100 and 101: A few species (e.g. Epinephelus gut
- Page 102 and 103: (a)(b)Fig. 1. Map of (a) study site
- Page 104 and 105: Fig. 2. Number of E. lanceolatus ob
- Page 106 and 107: was having any impact on the popula
- Page 108 and 109: A spawning aggregation is said to o
- Page 110 and 111: Chapter 11: Evaluation of an indica
- Page 112 and 113: Table 1 Aggregation fisheries asses
- Page 114 and 115: the lists of Jennings et al. (1999)
- Page 116 and 117: with the more vulnerable labrids an
- Page 118 and 119: The remaining serranid populations
- Page 120 and 121: Spawning aggregation behaviour is c
- Page 122 and 123: tiger grouper, Mycteroperca tigris:
- Page 124 and 125: of protecting the normal residence
- Page 126 and 127: • Since grouper males are afforde
- Page 130 and 131: The approaches identified above are
- Page 132 and 133: during full moon periods. Siganus s
- Page 134 and 135: model, many parameter estimates are
- Page 136 and 137: ReferencesAbunge C (2011) Managing
- Page 138 and 139: Cox DR (1972) Regression models and
- Page 140 and 141: Grüss A, Kaplan DM, Hart DR (2011b
- Page 142 and 143: Kaunda-Arara B, Rose GA (2004a) Eff
- Page 144: Newcomer RT, Taylor DH, Guttman SI
- Page 147 and 148: Sancho G, Petersen CW, Lobel PS (20
- Page 149 and 150: Appendix 1. QuestionnaireMASMA SPAW
- Page 151 and 152: 8. Spawning aggregation knowledgeUs
- Page 153 and 154: Example items KSh Furthest site Clo
- Page 155 and 156: Appendix II. Experimental testing o
- Page 157 and 158: Clove oil concentrationAt a concent
- Page 159 and 160: Appendix III. Application of acoust
- Page 161 and 162: 153