Зарегистрировано в Минюсте РФ 22 марта 2002 г
Зарегистрировано в Минюсте РФ 22 марта 2002 г
Зарегистрировано в Минюсте РФ 22 марта 2002 г
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
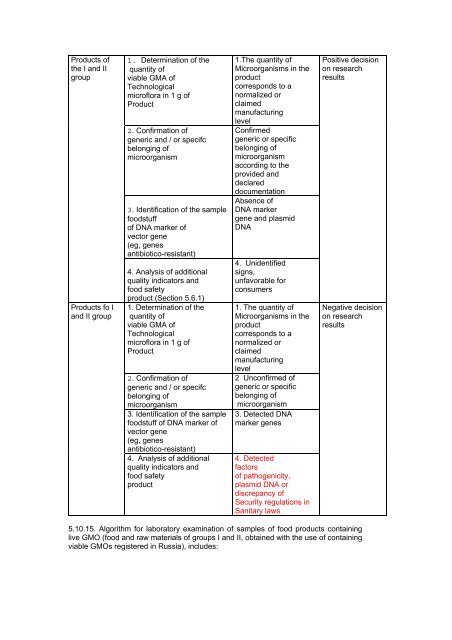
Products of<br />
the I and II<br />
group<br />
Products fo I<br />
and II group<br />
1. Determination of the<br />
quantity of<br />
viable GMA of<br />
Technological<br />
microflora in 1 g of<br />
Product<br />
2. Confirmation of<br />
generic and / or specifc<br />
belonging of<br />
microorganism<br />
3. Identification of the sample<br />
foodstuff<br />
of DNA marker of<br />
vector gene<br />
(eg, genes<br />
antibiotico-resistant)<br />
4. Analysis of additional<br />
quality indicators and<br />
food safety<br />
product (Section 5.6.1)<br />
1. Determination of the<br />
quantity of<br />
viable GMA of<br />
Technological<br />
microflora in 1 g of<br />
Product<br />
2. Confirmation of<br />
generic and / or specifc<br />
belonging of<br />
microorganism<br />
3. Identification of the sample<br />
foodstuff of DNA marker of<br />
vector gene<br />
(eg, genes<br />
antibiotico-resistant)<br />
4. Analysis of additional<br />
quality indicators and<br />
food safety<br />
product<br />
1.The quantity of<br />
Microorganisms in the<br />
product<br />
corresponds to a<br />
normalized or<br />
claimed<br />
manufacturing<br />
level<br />
Confirmed<br />
generic or specific<br />
belonging of<br />
microorganism<br />
according to the<br />
provided and<br />
declared<br />
documentation<br />
Absence of<br />
DNA marker<br />
gene and plasmid<br />
DNA<br />
4. Unidentified<br />
signs,<br />
unfavorable for<br />
consumers<br />
1. The quantity of<br />
Microorganisms in the<br />
product<br />
corresponds to a<br />
normalized or<br />
claimed<br />
manufacturing<br />
level<br />
2 Unconfirmed of<br />
generic or specific<br />
belonging of<br />
microorganism<br />
3. Detected DNA<br />
marker genes<br />
4. Detected<br />
factors<br />
of pathogenicity,<br />
plasmid DNA or<br />
discrepancy of<br />
Security regulations in<br />
Sanitary laws<br />
Positive decision<br />
on research<br />
results<br />
Negative decision<br />
on research<br />
results<br />
5.10.15. Algorithm for laboratory examination of samples of food products containing<br />
live GMO (food and raw materials of groups I and II, obtained with the use of containing<br />
viable GMOs registered in Russia), includes: