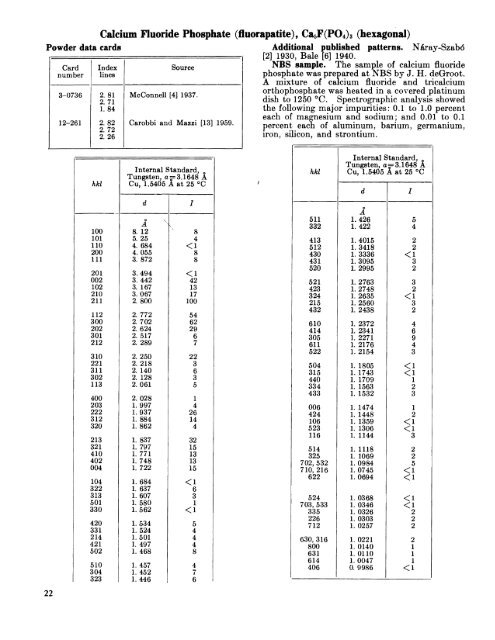
<strong>Powder</strong> data cards22Cardnumber3-073612-261hkl100101110200111201002102210211112300202301212310221311302113400203222312320213321410402004104322313501330420331214421502510304323Calcium Fluoride Phosphate (fluorapatite), Ca*F(PO4)s (hexagonal)Indexlines2. 812.711.842. 822.722.26McConnell [4] 1937.Carobbi and Mazzi [13] 1959.Internal <strong>Standard</strong>,Tungsten, a ==3. 1648 ACu, 1.5405 A at 25 °CdA v8. 125, 254. 6844.0553. 8723.4943.4423. 1673.0672. 8002. 7722. 7022. 6242.5172. 2892. 2502. 2182. 1402. 1282.0612. 0281.9971.9371. 8841. 8621. 8371. 7971. 7711. 7481. 7221. 6841. 6371.6071. 5801.5621.5341.5241.5011. 4971.4681.4571. 4521.446Additional published patterns. Nd<strong>ray</strong>-Szab6[2] 1930, Bale [6] 1940.SourceNBS sample* The sample of calcium fluoridephosphate was prepared at NBS by J. H. deGroot.A mixture of calcium fluoride and tricalciumorthophosphate was heated in a covered platinumdish to 1250 °C. Spectrographic analysis showedthe following major impurities: 0.1 to 1.0 percenteach of magnesium and sodium; and 0.01 to 0.1percent each of aluminum, barium, germanium,iron, silicon, and strontium.Internal <strong>Standard</strong>,Tungsten, a =3. 1648 Ahkl Cu, 1.5405 A at 25 Cd/IA511 1.42653321. 4224844131. 40152
Calcium Fluoride Phosphate (fluorapatite), Ca5F(PO4) 3 (hexagonal)—ContinuedThe sample was colorless and optically negativewith the indices of refraction N0= 1.633 and N e= 1.628.The d-values of the three strongest lines are:2.800, 2.702, and 2.772 A.Structural data. Hentschel [1] in 1923 determined that fluorapatite has the space groupC§h-P6/m (No. 176) and 2[Ca5F(PO4 ) 3] per unitcell.The density of synthetic fluorapatite calculatedfrom the NBS lattice constants is 3.201 g/cm 3 at25 °C.Lattice constantsac1923193019311939194019411946195019521952195219591963Hentschel [1]— ______-___-___-„__-----_-Nd<strong>ray</strong>-Szab6 [2]. ._.._..._______.._..____..Mehmel[3]_. _______„.___.____._.___._____Thewlis, Clock, and Mur<strong>ray</strong> [5] ,...._ __._..Bale [6]_ -----_-_------__----------------Klement and Dihn [?]___„. ________________Beevers and Maclntyre [8]_ _ ___ -Wallaeys and Chaudron [9] _ _ _ _ _Brasseur [10]__ _ _______ ___ ______ _ _McConnell [ll]________.__________-___-___-Altschuler, Cisney and Barlow [12] _ _____Carobbi and Mazzi [13]National Bureau of <strong>Standard</strong>s synthetic sample described above.Sample from Durango, Mex, a _ _ _Sample from Llallagua, Bolivia. b -_ __ .. _A/9. 33\9. 439.399. 389.389. 399. 389. 399. 3509. 3649. 3959. 3869. 424*9. 36849. 39239. 3712A6. 836. 896. 896. 866. 886. 896. 896. 896. 8706. 8796. 8826.8786. 888*6. 8841 at 25 °C6. 8821 at 25 °C6. 8824 at 26 °C*The error in these values was ±0.0003.••b These samples were obtained from the National Museum; the number of the Mexican sample is 104021, of the Bolivian sample 103869. These cell constants were derived from powder diffraction measurements made at the same time as those on the synthetic sample.» This particular sample from Durango may not be typical because of its lanthanum content. Spectropraphic analysis showed 1 to 4 percent of lanthanumand 0.1 to 1.0 percent each of cerium and sodium as major impurities. The error on these lattice constants was ±0.0005.b Spectrographic analysis showed major impurities to be 0,1 to 1.0 percent of manganese and 0.01 to 0.1 percent each of iron and strontium.References[1] H. Hentschel, Rontgenographische Untersuchungenam Apatit, Centr. Mineral. Geol. p. 609 (1923).[2] St. Na<strong>ray</strong>-Szab6, The structure of apatite (CaF)Ca4(PO 4) a , Z- Krist. 75, 387-398 (1930).[3] M. Mehmel, Beziehungen zwischen kristallstrukturund chemischer form el des apatits, Z. physik Chem.B15, 223-41 (1931).[4] D. McConnell, The substitution of SiO 4- and SO4-groups for PO4- groups in the apatite structure;ellestadite, the end-member, Am. Mineralogist 22,981 (1937).[5] J. Thewlis, G. E. Glock, and M. M. Mur<strong>ray</strong>, Chemicaland x-Ray analysis of dental, mineral, and syntheticapatites, Trans. Faraday Soc. 35, 358-63 (1939).[6] W. F. Bale, A comparative roentgen-<strong>ray</strong> diffractionstudy of several natural apatites and the apatitelikeconstituent of bone and tooth substance, Am.J. Roentgenol. 43, 735-47 (1940).[7] R. Klement and P. Dihn, Isomorphe Apatitarten,Naturwissenschaften 29, 301 (1941).[8] C. A. Beevers and D. B. Maclntyre, The atomicstructure of fluorapatite and its relation to that oftooth and bone material, Mineralog. Mag. 27, 254(1946).[9] R. Wallaeys and G. Chaudron, Sur la preparation decertaines apatites inixtes, Compt. rend. 231, 355-357 (1950).[10] H. Brasseur, Note sur les constitutes reticulaires etles indices de refraction des ftuor-, chlor-, et hydroxylapatites,Proceedings of the InternationalSymposium on the reactivity of solids, Gothei.burgPart 1, 363-7 (1952).[11] D. McConnell, The Problem of the carbonate apatitesIV, Structural substitutions involving CO 3 and OH,Bull. Soc. Franc. Mineral, et Crist. 75, 428 (1952).[12] Z. S. Altschuler, E. A. Cisney, and I. II. Barlow,X-Ray evidence of the nature of carbonate-apatite,Bull. Geol. Soc. Amer. 63, 1230-31 (1952).[13] G. Carobbi and F. Mazzi, Sulla possibilitu di unasostituzione parziale del calcio con 1'uranio nelreticolo dell' apatite, Atti. accad. naz. Lincei, Mem.Classe Sci. Fis. Mat. Nut. Ser. II* [8] 5, 159-71(1959).23
- Page 1 and 2: c ^r.
- Page 3 and 4: UNITED STATES DEPARTMENT OF COMMERC
- Page 5 and 6: Introduction.. _ __________________
- Page 7 and 8: STANDARD X-RAY DIFFRACTION POWDER P
- Page 9 and 10: D. E. Appleman, and D. Handworker.
- Page 11 and 12: Ammonium Fluoberyllate, (NH4) 2BeF4
- Page 13 and 14: Ammonium Fluoborate, NH4BF4 (orthor
- Page 15 and 16: Arsenic Trioxide, claudetite, As2O3
- Page 17 and 18: Powder data cardsBarium Stannate, B
- Page 19 and 20: Bismuth Orthophosphate, BiPO4 (trig
- Page 21 and 22: Bismuth Orthovanadate (high form),
- Page 23 and 24: Bismuth Telluride (tellurobismuthit
- Page 25 and 26: Cadmium Perchlorate Hexahydrate, Cd
- Page 27: Cadmium Telluride, CdTe (cubic)Powd
- Page 31 and 32: Cesium Chromate, Cs>CrO 4 (orthorho
- Page 33 and 34: Cobalt Fluosilkate Hexahydrate, CoS
- Page 35 and 36: Copper Sulfate (chalcocyanite), CuS
- Page 37 and 38: Erbium Arsenate, ErAsO4 (tetragonal
- Page 39 and 40: Gallium Arsenide, GaAs (cubic)Powde
- Page 41 and 42: Indium Arsenide, InAs (cubic)Powder
- Page 43 and 44: Lanthanum Niobium Titanium Oxide, L
- Page 45 and 46: Lithium Phosphate, high form, Li3PO
- Page 47 and 48: Magnesium Ammonium Phosphate Hexahy
- Page 49 and 50: Potossium Chlorate, KCIO3 (monoclin
- Page 51 and 52: Potassium Zinc Decavanadate 16 Hydr
- Page 53 and 54: Silver Antimony Telluride, AgSbTe2
- Page 55 and 56: Sodium Trimetaphosphate, Na3P3O9 (o
- Page 57 and 58: Sodium Trimetaphosphate Monohydrate
- Page 59 and 60: Strontium 1:1 Berate, SrO-B2O3 (ort
- Page 61 and 62: Thallium Chromate, Tl2CrO4 (orthorh
- Page 63 and 64: Titanium Dioxide, brookite, TiO2 (o
- Page 65 and 66: CUMULATIVE INDEX TO CIRCULAR 539, V
- Page 67 and 68: CUMULATIVE INDEX TO CIRCULAR 539, V
- Page 69 and 70: CUMULATIVE INDEX TO CIRCULAR 539, V