Lab Manual - eScience Labs
Lab Manual - eScience Labs
Lab Manual - eScience Labs
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Lab</strong> 5: Chemistry of Life<br />
Concepts to explore:<br />
• Atoms<br />
• Elements<br />
• Compounds<br />
• Chemical bonds<br />
• Molecules/Macromolecules<br />
• Energy and metabolism<br />
Concepts to explore:<br />
• Acids and bases<br />
• The effects of surface area<br />
and volume<br />
Introducon<br />
It is important to have a general understanding of chemistry<br />
before you can begin to understand how living organisms manage<br />
to reproduce, grow, move, eat, and perform a great many<br />
more funcons. To begin understanding the myriad of reac-<br />
Remember: Mass is the quanty of<br />
maer an object has; weight is the<br />
force produced by gravity acng on<br />
the mass of an object<br />
ons that occur within a cell, it is important to review the basics of chemistry. Recall that anything that<br />
occupies space and has mass is called maer; all maer is made of atoms.<br />
Atoms are made of a nucleus and two kinds of subatomic parcles: electrons (negavely charged parcles),<br />
and protons (posively charged parcles). Elements are pure substances that are made of only<br />
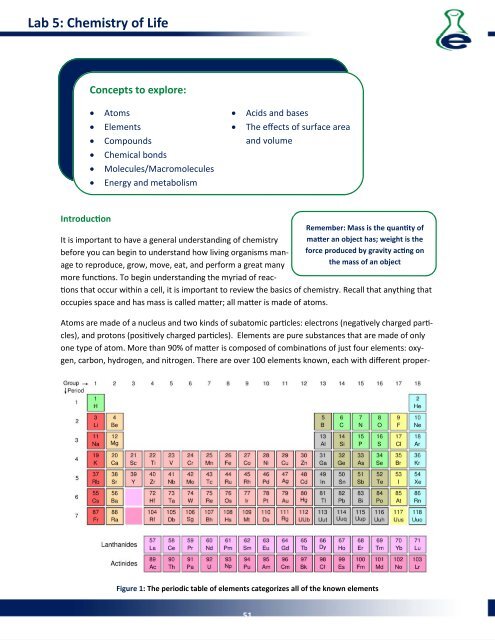
one type of atom. More than 90% of maer is composed of combinaons of just four elements: oxygen,<br />
carbon, hydrogen, and nitrogen. There are over 100 elements known, each with different proper-<br />
Figure 1: The periodic table of elements categorizes all of the known elements<br />
51