Human Rights and HIV/AIDSparticular, to promote access to medicinesfor all.”While the Doha Declaration was primarilya political statement, it lays down clearlegal guidelines for dealing with cases inthe dispute settlement process, and is animportant and necessary reaffirmation ofthe precedence of public health concernsover patents. Since the Declaration,however, WTO members have found itdifficult to reach an agreement on ways ofoperationalising it. Following thediscussions at the mini-ministerial inSydney, Australia, in 2002, the chairmanof the WTO TRIPS Council released a drafton 19 November 2002 for a decision bythe General Council. Most countriesagreed with the chair’s proposal of amoratorium on disputes regarding TRIPStill the Agreement is amended. However,disagreements remain over certainelements of the solution such as coverage,scope of eligible countries, etc.The Doha Declaration was particularlyimportant for the South <strong>Asia</strong>n region,because most countries are dependent onimported HIV/AIDS drugs, and, giventhe low purchasing power in thesecountries, the cost of drugs is an importantconcern in the provision of treatment.However, the Declaration and theflexibilities in TRIPS at the internationallevel will only partially help in ensuringaccess. Eventually, implementation ofthese provisions will be done throughnational legislation and appropriatedomestic policy.Most WTO members in the South <strong>Asia</strong>nregion have already altered their patentlaws to make them TRIPS-compliant.India, Pakistan and Sri Lanka have timeuntil 1 January 2005 before bringing thepharmaceutical sector under TRIPS.However, it is not clear if all theamendments carried out fully takeadvantage of the flexibilities under theBox 4.15Clarifications under the Doha DeclarationArticle 31: The TRIPS Agreement, under Article 31,allows for compulsory licensing under a set of conditions.The Doha Declaration increases the flexibility availableto member-countries to define the circumstances underwhich compulsory licenses are necessary. Under TRIPS,governments are expected to negotiate with patentholders for voluntary licenses on reasonable terms beforeissuing compulsory licenses, except in the case ofnational emergencies. The Doha Declaration leaves thedefinition and scope of national emergencies to thegovernments, giving them the discretion to categorisepublic health crises as national emergencies if necessary.Article 31(f) of the TRIPS Agreement states thatcompulsory licenses should be issued primarily forsupply in the domestic market. The Doha Declarationrecognises that this does not address the problem ofcountries with little or no manufacturing capacity, orwith insufficient domestic demand that are completelydependent on imports. Several proposals are currentlyunder discussion at the WTO to identify potentialoptions for imports of generic drugs.Article 30: This section provides for limited exceptionsto exclusive rights 67 provided under patents. Severalcountries and experts have proposed an authoritativeinterpretation under Article 30 as a solution to theproblem of allowing generic drugs for exports 68 thatis more streamlined and administratively simpler toexecute than Article 31.Article 6: The TRIPS Agreement leaves countries freeto decide the principle of exhaustion of IP rights thatthey wish to follow, subject to national treatment andmost favoured nations principles. The principle ofexhaustion defines the point of sale at which theproducer’s right over the product expires. The DohaDeclaration reaffirmed the right of countries topractise the principle of international exhaustion ofIP laws, allowing for private purchase and resale ofpatented products across countries through parallelimports. While this is an important provision for privatesuppliers of drugs, it does not resolve the problem oflarge-scale purchase of drugs at affordable rates.<strong>Regional</strong> Human Development <strong>Report</strong>HIV/AIDS and Development in South <strong>Asia</strong> 2003 103
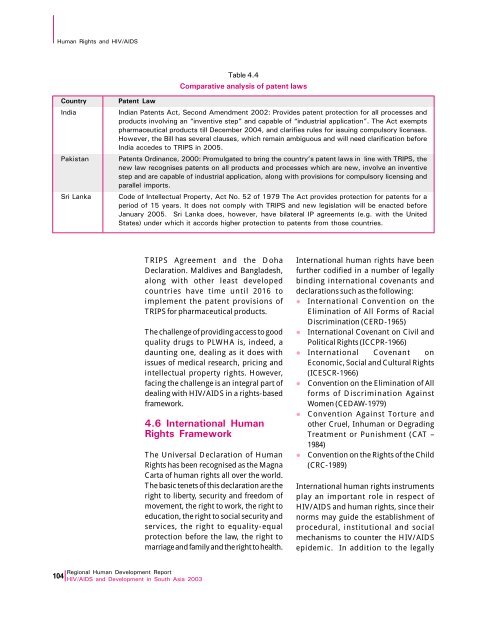
Human Rights and HIV/AIDSTable 4.4Comparative analysis of patent lawsCountryIndiaPakistanSri LankaPatent LawIndian Patents Act, Second Amendment 2002: Provides patent protection for all processes andproducts involving an “inventive step” and capable of “industrial application”. The Act exemptspharmaceutical products till December 2004, and clarifies rules for issuing compulsory licenses.However, the Bill has several clauses, which remain ambiguous and will need clarification beforeIndia accedes to TRIPS in 2005.Patents Ordinance, 2000: Promulgated to bring the country’s patent laws in line with TRIPS, thenew law recognises patents on all products and processes which are new, involve an inventivestep and are capable of industrial application, along with provisions for compulsory licensing andparallel imports.Code of Intellectual Property, Act No. 52 of 1979 The Act provides protection for patents for aperiod of 15 years. It does not comply with TRIPS and new legislation will be enacted beforeJanuary 2005. Sri Lanka does, however, have bilateral IP agreements (e.g. with the <strong>United</strong>States) under which it accords higher protection to patents from those countries.TRIPS Agreement and the DohaDeclaration. Maldives and Bangladesh,along with other least developedcountries have time until 2016 toimplement the patent provisions ofTRIPS for pharmaceutical products.The challenge of providing access to goodquality drugs to PLWHA is, indeed, adaunting one, dealing as it does withissues of medical research, pricing andintellectual property rights. However,facing the challenge is an integral part ofdealing with HIV/AIDS in a rights-basedframework.4.6 International HumanRights rameworkThe Universal Declaration of HumanRights has been recognised as the MagnaCarta of human rights all over the world.The basic tenets of this declaration are theright to liberty, security and freedom ofmovement, the right to work, the right toeducation, the right to social security andservices, the right to equality-equalprotection before the law, the right tomarriage and family and the right to health.International human rights have beenfurther codified in a number of legallybinding international covenants anddeclarations such as the following:l International Convention on theElimination of All Forms of RacialDiscrimination (CERD-1965)l International Covenant on Civil andPolitical Rights (ICCPR-1966)l International Covenant onEconomic, Social and Cultural Rights(ICESCR-1966)l Convention on the Elimination of Allforms of Discrimination AgainstWomen (CEDAW-1979)l Convention Against Torture andother Cruel, Inhuman or DegradingTreatment or Punishment (CAT –1984)l Convention on the Rights of the Child(CRC-1989)International human rights instrumentsplay an important role in respect ofHIV/AIDS and human rights, since theirnorms may guide the establishment ofprocedural, institutional and socialmechanisms to counter the HIV/AIDSepidemic. In addition to the legally<strong>Regional</strong> Human Development <strong>Report</strong>104 HIV/AIDS and Development in South <strong>Asia</strong> 2003