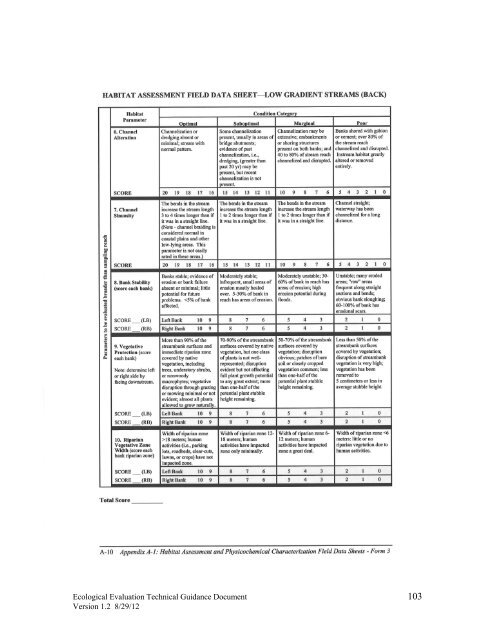
<strong>Ecological</strong> <strong>Evaluation</strong> <strong>Technical</strong> <strong>Guidance</strong> Document 103Version 1.2 8/29/12
Appendix B - Sampling Procedures for Benthic Algae and PlanktonAlgaeBenthic algae (periphyton) are primary producers and important foundation <strong>of</strong> manystream food webs. Periphyton also stabilize substrata and serves as habitat for many otherorganisms. Their characteristics are affected by physical, chemical, and biologicaldisturbances that may occur in the stream reach.Establish the sampling reach. Collect samples using techniques for specific substratetypes.Removable substrates (hard): gravel, pebbles, cobble, and woody debris. – Removerepresentative substrates from the water; brush or scrape a representative area <strong>of</strong> algaefrom the surface and rinse into a sample jar.Removable substrates (s<strong>of</strong>t): mosses, macroalgae, vascular plants, root masses. –Place a portion <strong>of</strong> the plant in a sample container with some water. Shake it vigorouslyand rub it gently to remove algae. Remove the plant from sample container.Large substrates (not removable): boulders, bedrock, logs, trees, and roots. - PlacePVC pipe with a neoprene collar at one end on the substrate so that the collar is sealedagainst the substrate. Dislodge algae in the pipe with a toothbrush or scraper. Removealgae from the pipe with pipette.Loose sediments: sand, silt, fine particulate organic matter, clay. – Invert a petri dishover sediments. Trap sediments in the petri dish by inserting a spatula under the dish.Remove sediment from the stream and rinse it into the sampling container. Algal samplesfrom depositional habitats can also be collected with spoons, forceps, or pipette. Placesamples collected from all substrate types into a single watertight, unbreakable, widemouthcontainer. If a single habitat is sampled, collect from several areas. A compositesample measuring four ounces (125 ml) is sufficient. Add preservative, and place a labelwith pertinent information on the outside <strong>of</strong> the container.Samples should be preserved (Lugol’s solution, 4% buffered formalin, “M3” fixative, or2% glutaraldehyde) and transported on ice and in the dark.ZooplanktonLocate sampling stations as near as possible to those stations selected for chemicalsampling to ensure maximum correlation <strong>of</strong> findings. These locations will depend uponthe physical nature <strong>of</strong> the water body. Stations should also be set up on either side <strong>of</strong> theriver to account for unequal lateral mixing. Slow-moving sections <strong>of</strong> streams generallycontain more zooplankton than slower-moving segments. If there are any lakes,reservoirs, or backwater areas upstream <strong>of</strong> sampling stations, notes on their nature andlocation should be included in the sampling log. Sampling stations in lakes, reservoirs,estuaries and the ocean should be located along grid networks or transect lines, aligned toprovide the most representative sampling. Points <strong>of</strong> interest should include intake anddischarge areas, constrictions within the water body, and major bays and tributaries <strong>of</strong>fthe main basin.Rivers, streams, shallow bays and coastal waters are usually well-mixed so that onlysubsurface sampling is necessary. In lakes and reservoirs, plankton composition and<strong>Ecological</strong> <strong>Evaluation</strong> <strong>Technical</strong> <strong>Guidance</strong> Document 104Version 1.2 8/29/12
- Page 1 and 2:
Ecological EvaluationTechnical Guid
- Page 3 and 4:
6.2.1.3 Biological Sampling of Fish
- Page 5 and 6:
Acronyms and AbbreviationsADDAETAFA
- Page 7 and 8:
Executive SummaryThis document prov
- Page 9 and 10:
environmentally sensitive areas pur
- Page 11 and 12:
Figure 3-1: Flow diagram to describ
- Page 13 and 14:
assessment may also include evaluat
- Page 15 and 16:
“Hazard quotient” or “HQ” m
- Page 17 and 18:
“Site investigation” means the
- Page 19 and 20:
parameters as specified in ERAGS (i
- Page 21 and 22:
document otherwise). The investigat
- Page 23 and 24:
5.3.2.1 Potential Contaminant Migra
- Page 25 and 26:
71 0Sampling pointsSampling transec
- Page 27 and 28:
5.3.4 Background ConsiderationsIt i
- Page 29 and 30:
III. GroundwaterAnalytical data fro
- Page 31 and 32:
5.5 Ecological Evaluation ReportThe
- Page 33 and 34:
Step 1 - Preliminary Screening Leve
- Page 35 and 36:
specific measurements of receptor h
- Page 37 and 38:
Figure 6-2: Ecological Conceptual S
- Page 39 and 40:
ingested, air inhaled, or material
- Page 41 and 42:
Fugacity, which is described as the
- Page 43 and 44:
environment. As noted in ERAGS, the
- Page 45 and 46:
Sample SelectionAfter completing th
- Page 47 and 48:
While there are many laboratories t
- Page 49 and 50:
ioavailability, and by doing so, of
- Page 51 and 52:
For the purposes of surface water,
- Page 53 and 54: higher trophic level receptors. Lip
- Page 55 and 56: Details regarding surface water tox
- Page 57 and 58: e present at intervals greater than
- Page 59 and 60: elatively sedentary organisms that
- Page 61 and 62: COPECs. The following references ar
- Page 63 and 64: tests (USEPA, 2002e). After collect
- Page 65 and 66: multiple reference area soils repre
- Page 67 and 68: In ERAs, tissue residue analyses ar
- Page 69 and 70: Objectives of the ERA: including a
- Page 71 and 72: evaluation might necessitate the co
- Page 73 and 74: N.J.A.C. 7:26E-4.8(c)1. The ERA may
- Page 75 and 76: sediment (i.e., that fraction that
- Page 77 and 78: Twelve dioxin-like PCB congeners ha
- Page 79 and 80: indicates burial of potential dioxi
- Page 81 and 82: of evidence for evaluating risk unt
- Page 83 and 84: 7.2.1 Apparent Effects Threshold Ap
- Page 85 and 86: when site conditions are most simil
- Page 87 and 88: destroying 10 acres of the mature f
- Page 89 and 90: ASTM (American Society for Testing
- Page 91 and 92: Establishing Sediment Quality Crite
- Page 93 and 94: N.J.A.C. (New Jersey Administrative
- Page 95 and 96: USEPA. 1989c. Risk Assessment Guida
- Page 97 and 98: http://www.epa.gov/owow/oceans/regu
- Page 99 and 100: USEPA 2006a. Data Quality Assessmen
- Page 101 and 102: Appendix A - Habitat Survey FormsEc
- Page 103: Ecological Evaluation Technical Gui
- Page 107 and 108: Appendix C - Surface Water Toxicity
- Page 109 and 110: Short-term chronic studies, endpoin
- Page 111 and 112: Appendix D - Sediment Toxicity Test
- Page 113 and 114: Toxicity Test DesignSediment toxici
- Page 115 and 116: Appendix E - Sediment Pore Water an
- Page 117 and 118: The seven-day daphnid survival and
- Page 119 and 120: esults are then evaluated using USE
- Page 121 and 122: Surber or Square-foot BottomThis sa
- Page 123 and 124: Appendix H - Soil Toxicity TestingS
- Page 125 and 126: another sample may still have a sub
- Page 127 and 128: conservative approach from an ecolo
- Page 129: Data PresentationTabular presentati