Role of the ubiquitin-like modifier FAT10 in protein degradation and ...
Role of the ubiquitin-like modifier FAT10 in protein degradation and ...
Role of the ubiquitin-like modifier FAT10 in protein degradation and ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Chapter 5<br />
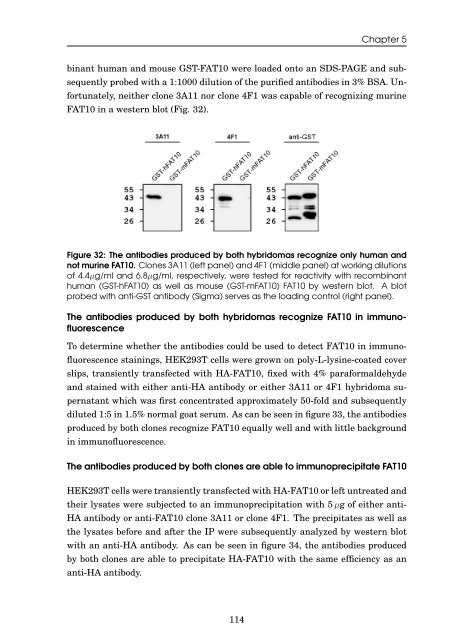
b<strong>in</strong>ant human <strong>and</strong> mouse GST-<strong>FAT10</strong> were loaded onto an SDS-PAGE <strong>and</strong> sub-<br />
sequently probed with a 1:1000 dilution <strong>of</strong> <strong>the</strong> purified antibodies <strong>in</strong> 3% BSA. Un-<br />
fortunately, nei<strong>the</strong>r clone 3A11 nor clone 4F1 was capable <strong>of</strong> recogniz<strong>in</strong>g mur<strong>in</strong>e<br />
<strong>FAT10</strong> <strong>in</strong> a western blot (Fig. 32).<br />
Figure 32: The antibodies produced by both hybridomas recognize only human <strong>and</strong><br />
not mur<strong>in</strong>e <strong>FAT10</strong>. Clones 3A11 (left panel) <strong>and</strong> 4F1 (middle panel) at work<strong>in</strong>g dilutions<br />
<strong>of</strong> 4.4µg/ml <strong>and</strong> 6.8µg/ml, respectively, were tested for reactivity with recomb<strong>in</strong>ant<br />
human (GST-h<strong>FAT10</strong>) as well as mouse (GST-m<strong>FAT10</strong>) <strong>FAT10</strong> by western blot. A blot<br />
probed with anti-GST antibody (Sigma) serves as <strong>the</strong> load<strong>in</strong>g control (right panel).<br />
The antibodies produced by both hybridomas recognize <strong>FAT10</strong> <strong>in</strong> immun<strong>of</strong>luorescence<br />
To determ<strong>in</strong>e whe<strong>the</strong>r <strong>the</strong> antibodies could be used to detect <strong>FAT10</strong> <strong>in</strong> immuno-<br />
fluorescence sta<strong>in</strong><strong>in</strong>gs, HEK293T cells were grown on poly-L-lys<strong>in</strong>e-coated cover<br />
slips, transiently transfected with HA-<strong>FAT10</strong>, fixed with 4% paraformaldehyde<br />
<strong>and</strong> sta<strong>in</strong>ed with ei<strong>the</strong>r anti-HA antibody or ei<strong>the</strong>r 3A11 or 4F1 hybridoma su-<br />
pernatant which was first concentrated approximately 50-fold <strong>and</strong> subsequently<br />
diluted 1:5 <strong>in</strong> 1.5% normal goat serum. As can be seen <strong>in</strong> figure 33, <strong>the</strong> antibodies<br />
produced by both clones recognize <strong>FAT10</strong> equally well <strong>and</strong> with little background<br />
<strong>in</strong> immun<strong>of</strong>luorescence.<br />
The antibodies produced by both clones are able to immunoprecipitate <strong>FAT10</strong><br />
HEK293T cells were transiently transfected with HA-<strong>FAT10</strong> or left untreated <strong>and</strong><br />
<strong>the</strong>ir lysates were subjected to an immunoprecipitation with 5 µg <strong>of</strong> ei<strong>the</strong>r anti-<br />
HA antibody or anti-<strong>FAT10</strong> clone 3A11 or clone 4F1. The precipitates as well as<br />
<strong>the</strong> lysates before <strong>and</strong> after <strong>the</strong> IP were subsequently analyzed by western blot<br />
with an anti-HA antibody. As can be seen <strong>in</strong> figure 34, <strong>the</strong> antibodies produced<br />
by both clones are able to precipitate HA-<strong>FAT10</strong> with <strong>the</strong> same efficiency as an<br />
anti-HA antibody.<br />
114