download pdf - Institut für Umweltphysik - Ruprecht-Karls-Universität ...
download pdf - Institut für Umweltphysik - Ruprecht-Karls-Universität ...
download pdf - Institut für Umweltphysik - Ruprecht-Karls-Universität ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
2.2. STRATOSPHERIC RESEARCH GROUP 39<br />
2.2.1 Observational constraints on stratospheric ozone loss cycles<br />
André Butz (Marcel Dorf, Sebastian Kreycy, Lena Kritten, Cristina Prados, Benjamin Simmes,<br />
Frank Weidner, Klaus Pfeilsticker)<br />
Abstract Balloon-borne observations of a comprehensive set of trace gases in the Arctic winter<br />
stratosphere are used to constrain a photochemical model. Model-measurement comparisons aim<br />
at testing the consistency of various recently suggested scenarios of the involved reaction kinetics.<br />
Particular focus is put on the ClO-BrO and the ClO-ClO cycles, and on inferring implications for<br />
ozone loss.<br />
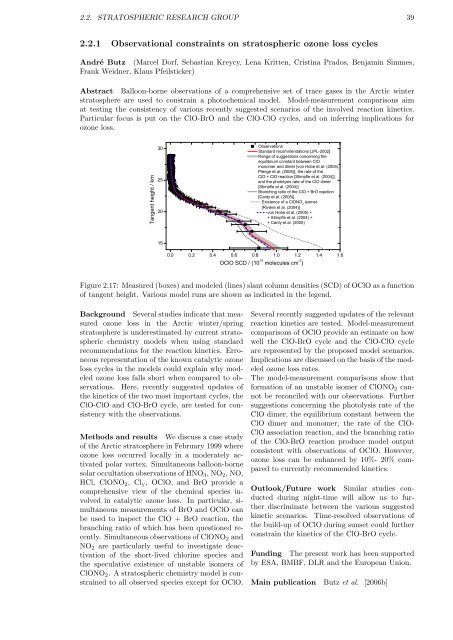
Figure 2.17: Measured (boxes) and modeled (lines) slant column densities (SCD) of OClO as a function<br />
of tangent height. Various model runs are shown as indicated in the legend.<br />
Background Several studies indicate that measured<br />
ozone loss in the Arctic winter/spring<br />
stratosphere is underestimated by current stratospheric<br />
chemistry models when using standard<br />
recommendations for the reaction kinetics. Erroneous<br />
representation of the known catalytic ozone<br />
loss cycles in the models could explain why modeled<br />
ozone loss falls short when compared to observations.<br />
Here, recently suggested updates of<br />
the kinetics of the two most important cycles, the<br />
ClO-ClO and ClO-BrO cycle, are tested for consistency<br />
with the observations.<br />
Methods and results We discuss a case study<br />
of the Arctic stratosphere in February 1999 where<br />
ozone loss occurred locally in a moderately activated<br />
polar vortex. Simultaneous balloon-borne<br />
solar occultation observations of HNO3, NO2, NO,<br />
HCl, ClONO2, Cly, OClO, and BrO provide a<br />
comprehensive view of the chemical species involved<br />
in catalytic ozone loss. In particular, simultaneous<br />
measurements of BrO and OClO can<br />
be used to inspect the ClO + BrO reaction, the<br />
branching ratio of which has been questioned recently.<br />
Simultaneous observations of ClONO2 and<br />
NO2 are particularly useful to investigate deactivation<br />
of the short-lived chlorine species and<br />
the speculative existence of unstable isomers of<br />
ClONO2. A stratospheric chemistry model is constrained<br />
to all observed species except for OClO.<br />
Several recently suggested updates of the relevant<br />
reaction kinetics are tested. Model-measurement<br />
comparisons of OClO provide an estimate on how<br />
well the ClO-BrO cycle and the ClO-ClO cycle<br />
are represented by the proposed model scenarios.<br />
Implications are discussed on the basis of the modeled<br />
ozone loss rates.<br />
The model-measurement comparisons show that<br />
formation of an unstable isomer of ClONO2 cannot<br />
be reconciled with our observations. Further<br />
suggestions concerning the photolysis rate of the<br />
ClO dimer, the equilibrium constant between the<br />
ClO dimer and monomer, the rate of the ClO-<br />
ClO association reaction, and the branching ratio<br />
of the ClO-BrO reaction produce model output<br />
consistent with observations of OClO. However,<br />
ozone loss can be enhanced by 10%- 20% compared<br />
to currently recommended kinetics.<br />
Outlook/Future work Similar studies conducted<br />
during night-time will allow us to further<br />
discriminate between the various suggested<br />
kinetic scenarios. Time-resolved observations of<br />
the build-up of OClO during sunset could further<br />
constrain the kinetics of the ClO-BrO cycle.<br />
Funding The present work has been supported<br />
by ESA, BMBF, DLR and the European Union.<br />
Main publication Butz et al. [2006b]