download pdf - Institut für Umweltphysik - Ruprecht-Karls-Universität ...
download pdf - Institut für Umweltphysik - Ruprecht-Karls-Universität ...
download pdf - Institut für Umweltphysik - Ruprecht-Karls-Universität ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
2.5. MARHAL - MODELING OF MARINE AND HALOGEN CHEMISTRY 77<br />
2.5.2 The Potential Importance of Frost Flowers for Ozone Depletion Events<br />
- A Model Study<br />
Matthias Piot, Roland von Glasow<br />
Abstract For more than 20 years, events with almost complete loss of ozone have been observed<br />
in the Arctic in spring. We performed model studies with the one-dimensional model MISTRA to<br />
investigate the potential role of frost flowers (FF) in this depletion of tropospheric ozone.<br />
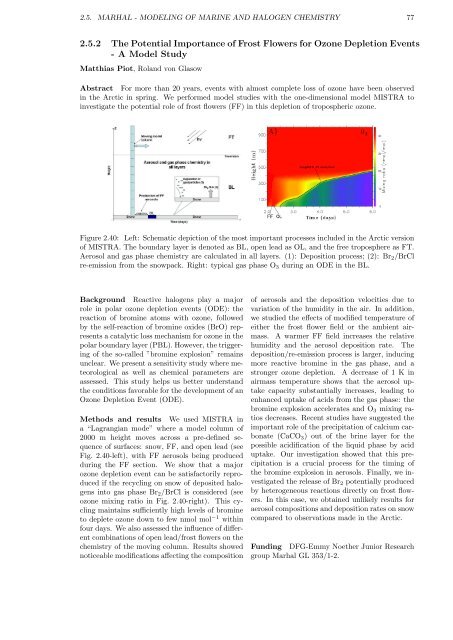
Figure 2.40: Left: Schematic depiction of the most important processes included in the Arctic version<br />
of MISTRA. The boundary layer is denoted as BL, open lead as OL, and the free troposphere as FT.<br />
Aerosol and gas phase chemistry are calculated in all layers. (1): Deposition process; (2): Br2/BrCl<br />
re-emission from the snowpack. Right: typical gas phase O3 during an ODE in the BL.<br />
Background Reactive halogens play a major<br />
role in polar ozone depletion events (ODE): the<br />
reaction of bromine atoms with ozone, followed<br />
by the self-reaction of bromine oxides (BrO) represents<br />
a catalytic loss mechanism for ozone in the<br />
polar boundary layer (PBL). However, the triggering<br />
of the so-called ”bromine explosion” remains<br />
unclear. We present a sensitivity study where meteorological<br />
as well as chemical parameters are<br />
assessed. This study helps us better understand<br />
the conditions favorable for the development of an<br />
Ozone Depletion Event (ODE).<br />
Methods and results We used MISTRA in<br />
a “Lagrangian mode” where a model column of<br />
2000 m height moves across a pre-defined sequence<br />
of surfaces: snow, FF, and open lead (see<br />
Fig. 2.40-left), with FF aerosols being produced<br />
during the FF section. We show that a major<br />
ozone depletion event can be satisfactorily reproduced<br />
if the recycling on snow of deposited halogens<br />
into gas phase Br2/BrCl is considered (see<br />
ozone mixing ratio in Fig. 2.40-right). This cycling<br />
maintains sufficiently high levels of bromine<br />
to deplete ozone down to few nmol mol −1 within<br />
four days. We also assessed the influence of different<br />
combinations of open lead/frost flowers on the<br />
chemistry of the moving column. Results showed<br />
noticeable modifications affecting the composition<br />
of aerosols and the deposition velocities due to<br />
variation of the humidity in the air. In addition,<br />
we studied the effects of modified temperature of<br />
either the frost flower field or the ambient airmass.<br />
A warmer FF field increases the relative<br />
humidity and the aerosol deposition rate. The<br />
deposition/re-emission process is larger, inducing<br />
more reactive bromine in the gas phase, and a<br />
stronger ozone depletion. A decrease of 1 K in<br />
airmass temperature shows that the aerosol uptake<br />
capacity substantially increases, leading to<br />
enhanced uptake of acids from the gas phase: the<br />
bromine explosion accelerates and O3 mixing ratios<br />
decreases. Recent studies have suggested the<br />
important role of the precipitation of calcium carbonate<br />
(CaCO3) out of the brine layer for the<br />
possible acidification of the liquid phase by acid<br />
uptake. Our investigation showed that this precipitation<br />
is a crucial process for the timing of<br />
the bromine explosion in aerosols. Finally, we investigated<br />
the release of Br2 potentially produced<br />
by heterogeneous reactions directly on frost flowers.<br />
In this case, we obtained unlikely results for<br />
aerosol compositions and deposition rates on snow<br />
compared to observations made in the Arctic.<br />
Funding DFG-Emmy Noether Junior Research<br />
group Marhal GL 353/1-2.