THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Di-acidic compounds. Mono-2- ethylhexylphosphoric acid has been most<br />
extensively studied of this type of compound. It is polymeric in the <strong>org</strong>amic phase in<br />
a non-polar solvent,<br />
such as ~enzene310, 130<br />
and extracts primarily by the ion exchange re-<br />
action.<br />
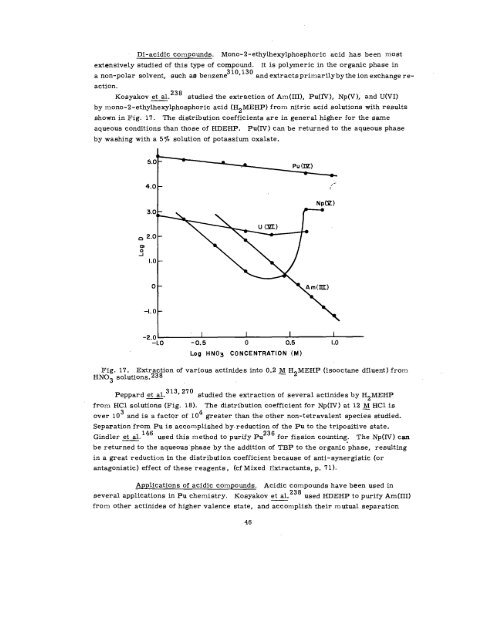
Kos yakov et al. 238 studied the extraction of Am(III), Pu(l!V), NP(V), and U(VI)<br />
by mono-2- ethylhexylphosphoric acid (H2MEHP) from nitric acid solutions with results<br />
shown in Fig. 17. The distribution coefficients are in general higher for the same<br />
aqueous conditions than those of HDEHP. Pu(IV) can be returned to the aqueous phase<br />
by washing with a 5 ~. solution of potassium oxalate.<br />
5.0 -<br />
4.0<br />
t<br />
~ 2.0 -<br />
:<br />
-1<br />
I.0 –<br />
o -<br />
-1.0 -<br />
NP(E)<br />
1 I I I<br />
-2.o~<br />
-1.0 -0.5 0 0.5 1.0<br />
Log HN03 CONCENTRATION (M)<br />
Fig. 17. Extraction of various actinides into 0.2 ~ H2MEHP (isooctane diluent ) from<br />
HN03 solutions.238<br />
Peppard et al, 313, 270 studied the extraction of several actinides by H2MEHP<br />
from HC1 solutions (Fig. 18). The distribution coefficient for Np(IV) at 12 ~ HC1 is<br />
over 103 and is a factor of 104 greater than the other non-tetravalent species studied.<br />
Separation from Pu is accomplished by. reduction of the Pu to the tripositive state.<br />
Gindler ~. 14’ used this method to purify<br />
236<br />
Pu for fission counting. The Np(IV) can<br />
be returned to the aqueous phase by the addition of TBP to the <strong>org</strong>anic phase, resulting<br />
in a great reduction in the distribution coefficient because of anti- synergistic (or<br />
antagonistic) effect of these reagents, (cf Mixed ExtractantsJ p. 71).<br />
Applications of acidic compounds. Acidic compounds have been used in<br />
several applications in Pu chemistry. Kosyakov et al. 238 used HDEHP to purify Am(lII)<br />
from other actinides of higher valence state, and accomplish their mutual separation<br />
46