THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
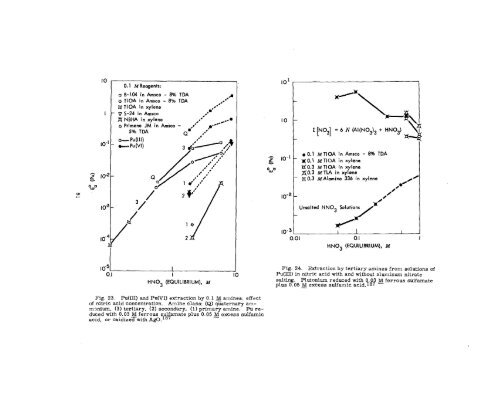
0.1 M Reogents:<br />
o B-lu in Amsco - w TDA ●*A<br />
o TIOA in Amsco - 8%<br />
M TI OA in xylene<br />
V S-24 in Amsco<br />
~ NBHA in xylene<br />
o Primene JM in Amsca<br />
TDA<br />
-<br />
.***<br />
.0<br />
■<br />
.“<br />
~..<br />
●’””<br />
,c* p--~<br />
●<br />
5% TDA<br />
Qm ●“<br />
I<br />
1’<br />
10<br />
2<br />
HN03 (EQUILIBRIUM), M<br />
Fig. 23. Pu(III) and Pu(VI) extraction by 0,1 ~ amines: effect<br />
of nitric acid concentration. Amine class: (Q) quaternary ammoniwn,<br />
(3) tertiary, (2) secondary, (1) primary amine. Pu reduced<br />
with 0.03 M ferrous su amate plus 0.05 ~ excess sulfamic<br />
acid, or oxidize~with AgO. 1 d{<br />
101<br />
10<br />
~ 10-1<br />
Ouo<br />
,0-2<br />
I<br />
~-~<br />
\<br />
Z N03 [1 = 6 IV (AI(N03)3 +<br />
–%<br />
HN03) \<br />
x-<br />
.0.1 MTIOA in Amsco - B% TDA<br />
xO. I MTIOA in xylena<br />
~0.3 MTIOA in xylene<br />
~1).3 M TLA in xylene<br />
- H 0.3 itfAlamine 336 in xylene /0~<br />
U~ltd HN03 Solutions<br />
/<br />
,0-3<br />
I I I<br />
0.01 0.1 I<br />
x’<br />
,0’<br />
HN03 (EQUILIBRIUM), M<br />
Fig. 24. Extraction by tertiary aminea from solutions of<br />
Pu(III) in nitric acid with and without aluminum nitrate<br />
Plutonium reduced with 0,0 ~ ferrouB sulf~ate<br />
~&%g05 M excess sulfamic acid, 18?<br />
—<br />
,’