THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
THE RADIOCHEMISTRY OF PLUTONIUM - Sciencemadness.org
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
‘“’m!r=% ‘“”EsFF?E<br />
D<br />
1.0<br />
lo-.17F%%F1<br />
I I I I 1 I<br />
I I [ 1 I<br />
,O-LLLLLLU<br />
0 2 4 6 B 1“ 12 14<br />
Aqueous HN03 Concentrotlon, ~<br />
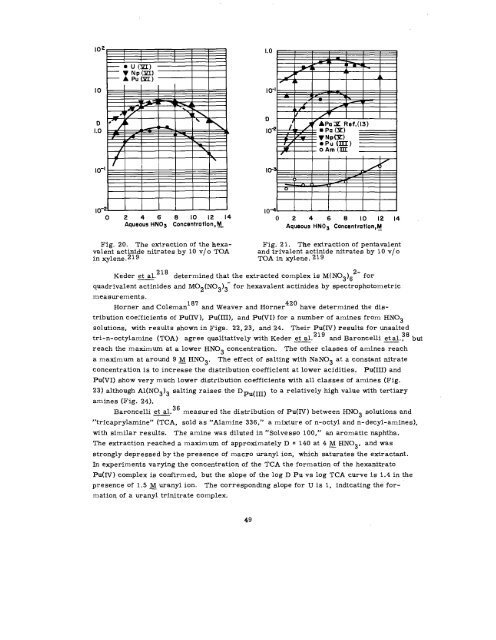
Fig. 20. The extraction of the hexavalent<br />
actinide nitrates by 10 v/o TOA<br />
in xylene.219<br />
Io+<br />
I I 1 I I I<br />
I f [ a<br />
AP I<br />
D<br />
,“* .<br />
1<br />
i ~ .x<br />
APa X Ref.(13)<br />
■ Pa (X)<br />
r A<br />
r~[<br />
* I<br />
/<br />
vNPCZ)<br />
● PU (m)<br />
o Am (~<br />
10-3<br />
7 I -<br />
0 I I<br />
I I<br />
O 2 4 6 8 10 12 14<br />
Aqueous HN03 Concentration, ~<br />
Fig. 21. The extraction of pentavalent<br />
and trivalent actinide nitrates by 10 v/o<br />
‘roA in xylene. 219<br />
Keder et al. 218 determined that the extracted complex is M(N03 )62- for<br />
quadrivalent actinides and M02 (N03 )3- for hexavalent actinides by spectrophotometric<br />
measurements.<br />
Homer and Coleman<br />
187 420<br />
and Weaver and Homer have determined the dis-<br />
tribution coefficients of Pu(IV), Pu(III), and Pu(VI) for a number of amines from HN03<br />
solutions, with results shown in Figs. 22, 23, and 24. Their Pu(IV) results for unsalted<br />
tri-n-octylamine (TOA ) agree qualitatively with Keder et al. 219 and Baroncelli ~.~’<br />
reach the maximum at a lower HN03 concentration. The other classes of amines reach<br />
a maximum at around 9 M HN03. The effect of salting with NaN03 at a constant nitrate<br />
—<br />
concentration is to increase the distribution coefficient at lower acidities. Pu(III) and<br />
Pu(VI) show very much lower distribution coefficients with all classes of amines (Fig.<br />
23) although A1(N03)3 salting raises the D ~u(lll) to a relatively high value with tertiary<br />
amines (Fig. 24).<br />
Baroncelli et al. 36 measured the distribution of Pu(17J) between HN03 solutions and<br />
“tricaprylamine” (TCA, sold as “Alamine 336, ” a mixture of n-octyl and n-decyl-amines),<br />
with similar results. The amine was diluted in “Solvesso 100, ” an aromatic naphtha.<br />
The extraction reached a maximum of approximately D = 140 at 4 ~ HN03, and was<br />
strongly depressed by the presence of macro uranyl ion, which saturates the extractant.<br />
In experiments varying the concentration of the TCA the formation of the hexanitrato<br />
Pu(IV) complex is confirmed, but the slope of the log D Pu vs log TCA curve is 1.4 in the<br />
presence of 1.5 ~ uranyl ion. The corresponding slope for U is 1, indicating the for-<br />
mation of a uranyl trinitrate complex.<br />
49<br />
but