Donepezil, rivastigmine, galantamine and memantine for ...
Donepezil, rivastigmine, galantamine and memantine for ...
Donepezil, rivastigmine, galantamine and memantine for ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
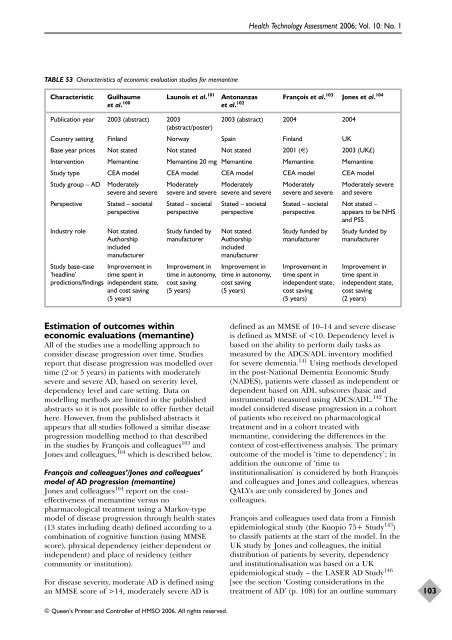
TABLE 53 Characteristics of economic evaluation studies <strong>for</strong> <strong>memantine</strong><br />
Estimation of outcomes within<br />
economic evaluations (<strong>memantine</strong>)<br />
All of the studies use a modelling approach to<br />
consider disease progression over time. Studies<br />
report that disease progression was modelled over<br />
time (2 or 5 years) in patients with moderately<br />
severe <strong>and</strong> severe AD, based on severity level,<br />
dependency level <strong>and</strong> care setting. Data on<br />
modelling methods are limited in the published<br />
abstracts so it is not possible to offer further detail<br />
here. However, from the published abstracts it<br />
appears that all studies followed a similar disease<br />
progression modelling method to that described<br />
in the studies by François <strong>and</strong> colleagues 103 <strong>and</strong><br />
Jones <strong>and</strong> colleagues, 104 which is described below.<br />
François <strong>and</strong> colleagues’/Jones <strong>and</strong> colleagues’<br />
model of AD progression (<strong>memantine</strong>)<br />
Jones <strong>and</strong> colleagues 104 report on the costeffectiveness<br />
of <strong>memantine</strong> versus no<br />
pharmacological treatment using a Markov-type<br />
model of disease progression through health states<br />
(13 states including death) defined according to a<br />
combination of cognitive function (using MMSE<br />
score), physical dependency (either dependent or<br />
independent) <strong>and</strong> place of residency (either<br />
community or institution).<br />
For disease severity, moderate AD is defined using<br />
an MMSE score of >14, moderately severe AD is<br />
© Queen’s Printer <strong>and</strong> Controller of HMSO 2006. All rights reserved.<br />
Health Technology Assessment 2006; Vol. 10: No. 1<br />
Characteristic Guilhaume Launois et al. 101 Antonanzas François et al. 103 Jones et al. 104<br />
et al. 100 et al. 102<br />
Publication year 2003 (abstract) 2003<br />
(abstract/poster)<br />
2003 (abstract) 2004 2004<br />
Country setting Finl<strong>and</strong> Norway Spain Finl<strong>and</strong> UK<br />
Base year prices Not stated Not stated Not stated 2001 (€) 2003 (UK£)<br />
Intervention Memantine Memantine 20 mg Memantine Memantine Memantine<br />
Study type CEA model CEA model CEA model CEA model CEA model<br />
Study group – AD Moderately Moderately Moderately Moderately Moderately severe<br />
severe <strong>and</strong> severe severe <strong>and</strong> severe severe <strong>and</strong> severe severe <strong>and</strong> severe <strong>and</strong> severe<br />
Perspective Stated – societal Stated – societal Stated – societal Stated – societal Not stated –<br />
perspective perspective perspective perspective appears to be NHS<br />
<strong>and</strong> PSS<br />
Industry role Not stated. Study funded by Not stated. Study funded by Study funded by<br />
Authorship manufacturer Authorship manufacturer manufacturer<br />
included included<br />
manufacturer manufacturer<br />
Study base-case Improvement in Improvement in Improvement in Improvement in Improvement in<br />
‘headline’ time spent in time in autonomy, time in autonomy, time spent in time spent in<br />
predictions/findings independent state, cost saving cost saving independent state, independent state,<br />
<strong>and</strong> cost saving (5 years) (5 years) cost saving cost saving<br />
(5 years) (5 years) (2 years)<br />
defined as an MMSE of 10–14 <strong>and</strong> severe disease<br />
is defined as MMSE of