CHEM01200604005 A. K. Pathak - Homi Bhabha National Institute
CHEM01200604005 A. K. Pathak - Homi Bhabha National Institute
CHEM01200604005 A. K. Pathak - Homi Bhabha National Institute
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
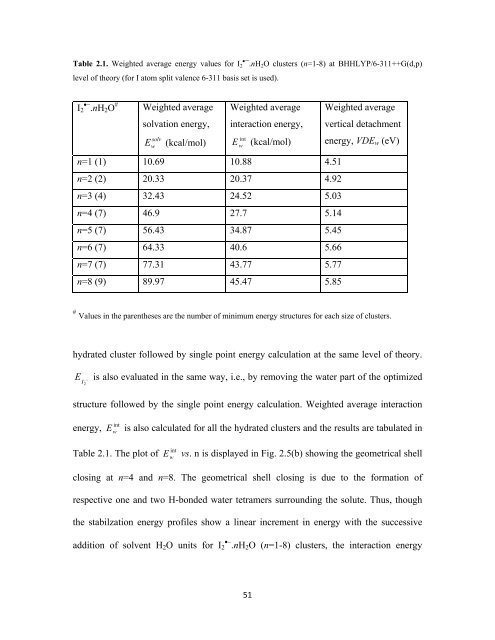
Table 2.1. Weighted average energy values for I 2•¯.nH 2 O clusters (n=1-8) at BHHLYP/6-311++G(d,p)<br />
level of theory (for I atom split valence 6-311 basis set is used).<br />
I 2 •− .nH 2 O #<br />
Weighted average<br />
Weighted average<br />
Weighted average<br />
solvation energy,<br />
interaction energy,<br />
vertical detachment<br />
E w solv (kcal/mol)<br />
E w<br />
int<br />
(kcal/mol)<br />
energy, VDE w (eV)<br />
n=1 (1) 10.69 10.88 4.51<br />
n=2 (2) 20.33 20.37 4.92<br />
n=3 (4) 32.43 24.52 5.03<br />
n=4 (7) 46.9 27.7 5.14<br />
n=5 (7) 56.43 34.87 5.45<br />
n=6 (7) 64.33 40.6 5.66<br />
n=7 (7) 77.31 43.77 5.77<br />
n=8 (9) 89.97 45.47 5.85<br />
# Values in the parentheses are the number of minimum energy structures for each size of clusters.<br />
hydrated cluster followed by single point energy calculation at the same level of theory.<br />
E<br />
I2<br />
.−<br />
is also evaluated in the same way, i.e., by removing the water part of the optimized<br />
structure followed by the single point energy calculation. Weighted average interaction<br />
energy,<br />
E w<br />
int<br />
is also calculated for all the hydrated clusters and the results are tabulated in<br />
Table 2.1. The plot of<br />
E w<br />
int<br />
vs. n is displayed in Fig. 2.5(b) showing the geometrical shell<br />
closing at n=4 and n=8. The geometrical shell closing is due to the formation of<br />
respective one and two H-bonded water tetramers surrounding the solute. Thus, though<br />
the stabilzation energy profiles show a linear increment in energy with the successive<br />
addition of solvent H 2 O units for I •− 2 .nH 2 O (n=1-8) clusters, the interaction energy<br />
51