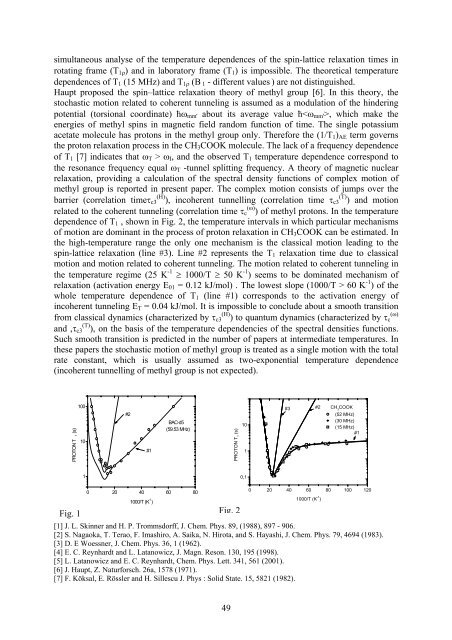
LOW TEMPERATURES DYNAMICS IN SYSTEMS CONSISTING OF HYDROGEN BOND AND METHYL GROUPS L. Latanowicz, W. Medycki*, J. Boguszyńska*, E. C. Reynhardt** Department of Biophysics, Institute of Biotechnology and Environmental Sciences, Zielona Góra University, Monte Cassino 21 B, 65-561 Zielona Gora, Poland; *Institute of Molecular Physics, Polish Academy of Sciences, Smoluchowskiego 17, 60-179 Poznań, Poland; **Department of Physics, University of South Africa, P O Box 392, Pretoria, 0003, SA The paper deals with the two different approaches to the spectral densities functions of proton transfer and methyl group rotation. The transfer of a hydrogen atom in a hydrogen bond is due to classical jumps over the potential barrier and incoherent tunnelling. The concerted jumps of two hydrogen bonded protons (or deuterons) along a hydrogen bond of benzoic acid can be visualised as an exchange motion between potential energy minima associated with tautomers A and B with different sets of rate constants, the first set obeying Arrhenius' law (rate constants of the jumps over the barrier) and the second set given by Skinner and Trommsdorff [1] or Nagaoka et al [2] (rate constants of the incoherent tunnelling). The preexponential factor of the rate of tunneling jumps (k tu 0 ) is usually in range 10 8 Hz for hydrogen (10 6 Hz for deuterium) while this of the rate of jumps over the barrier (k ov 0 ) is in range 10 12 Hz (10 10 Hz). If the interaction Hamiltonian is modulated by a series of independent stochastic processes, the total correlation function of such a complex motion should be calculated [3]. The equations for spectral densities functions of complex motion, which consists of jumps over the barrier and incoherent tunnelling were derived by Reynhardt and Latanowicz [4]. That the spectral density of incoherent tunnelling is eliminated above a certain temperature, leaving only the spectral density of the classical jump motion contributing to spin-lattice relaxation at higher temperatures has been revealed in the paper [5]. A comparison (Fig. 1) of measured (open circles [2]) and calculated T 1 (theory [4] - #1, jumps over the barrier - #2) for benzoic acid confirms this discovery. We have found that this event has justification in the quantum mechanics. Quantum mechanics involves probability of transfer of the particle through the barrier, which is serious different then probability of jumps over the barrier. This probability can be defined as D = I / I 0 , where I is intensity of de Broglie's wave (which describe the particle) outside of the potential wall and I 0 is this intensity inside the potential wall. The formula, which describes a coefficient D depends on the shape of the potential barrier. For the 2L rectangular barrier D = D exp[ − 2m( E E ) ] , where E AB and L are height and width 0 AB − of the barrier, E and m are energy and mass of the particle. The value (E AB - E) under square root is negative for E > E AB and the probability D is lost. The thermal energy of the Avogadro number of particles equals E = C p T [kJ/mol] where C p is molar heat capacity and T is temperature in the Kelvin scale. When C p T = E AB this should be the last value of coefficient D in the temperature dependence. In opposition to presented equations, the expression for spectral density functions of single motion with total rate constant, approximated by two exponents k AB = k ov tu 0 exp(-H/RT) + k 0 exp(-∆/RT) (H and ∆ are the high and low temperature slopes of T 1 ) has been used in number of papers. This last one theoretical approach assumes the smooth transition between the over the barrier rate constant and incoherent tunneling rate constant. Here the question appears how this is possible that the Arrhenius rate constant k ov = k 0 exp(-H/RT), goes to zero value above temperature zero Kelvin and classical motion ceases? Moreover, when "single motion approach" to the total spectral density functions is applied then T 1 looses the frequency dependence at minimum value for low resonance frequencies of spectrometer. The 48
simultaneous analyse of the temperature dependences of the spin-lattice relaxation times in rotating frame (T 1ρ ) and in laboratory frame (T 1 ) is impossible. The theoretical temperature dependences of T 1 (15 MHz) and T 1ρ (B 1 - different values ) are not distinguished. Haupt proposed the spin–lattice relaxation theory of methyl group [6]. In this theory, the stochastic motion related to coherent tunneling is assumed as a modulation of the hindering potential (torsional coordinate) ћω mm' about its average value ћ
- Page 1 and 2: The Henryk Niewodniczański INSTITU
- Page 3 and 4: Contents R. Banyś, A. Słowik, M.
- Page 5 and 6: J. Kolmas, M. Uniczko, E. Kalinowsk
- Page 7 and 8: Ł. Popenda, Z. Gdaniec, G. Dominia
- Page 9 and 10: USEFULNESS OF PROTON MAGNETIC RESON
- Page 11 and 12: INFLUENCE OF PARAMAGNETIC IONS ON F
- Page 13 and 14: NMR RELAXATION OF PAPER SAMPLES Bar
- Page 15 and 16: SPIN-LATTICE RELAXATION OF D 2 QUAN
- Page 17 and 18: INVESTIGATION OF NEUROPROTECTING EF
- Page 19 and 20: OPTICAL PUMPING OF 3 He Katarzyna C
- Page 21 and 22: NMR STUDY OF GELATION PROCESS OF LO
- Page 23 and 24: MRI INVESTIGATIONS OF HYDROGEL FORM
- Page 25 and 26: CHEMICAL SHIFT TITRATION AT THE INT
- Page 27 and 28: LOCAL DYNAMICS AND ORGANIZATION OF
- Page 29 and 30: 1 H NMR DETECTION OF σ-ADDUCTS IN
- Page 31 and 32: HYDRATION OF WHEAT PHOTOSYNTHETIC M
- Page 33 and 34: References: 1. H. Harańczyk On wat
- Page 35 and 36: (EDV) and end-systolic (ESV) volume
- Page 37 and 38: THE MOST STABLE POLYMORPHIC FORM OF
- Page 39 and 40: A DETERMINATION OF ABSOLUTE SHIELDI
- Page 41 and 42: DIAGNOTIC VALUE OF MRI IN CONVERSIO
- Page 43 and 44: NMR AND FTIR STUDY OF MOLECULAR DYN
- Page 45 and 46: EFFECTS OF INTERMOLECULAR INTERACTI
- Page 47: 600000 Integral of 1D MRI profils [
- Page 51 and 52: 1 H NMR STUDIES OF PLATINUM(II) CHL
- Page 53 and 54: 35 Cl-NQR STUDY OF ELECTRONIC STRUC
- Page 55 and 56: 35 Cl- NQR AND 1 H-NMR STUDY OF MOL
- Page 57 and 58: THE SYNTHESIS AND NMR ANALYSIS OF T
- Page 59 and 60: STUDY OF MOTIONAL PROCESSES OF DRAW
- Page 61 and 62: intensity (arb. units) 0,20 0,15 0,
- Page 63 and 64: data for Cβ-nonplanar model 3 are
- Page 65 and 66: T 1 DISPERSION AND SELF-DIFFUSION N
- Page 67 and 68: A LOW FIELD MRI SYSTEM FOR HYPERPOL
- Page 69 and 70: 1 H MAS NMR OF FLAVONOIDS Katarzyna
- Page 71 and 72: EFFECT OF TEMPERATURE ON LIPOSOME S
- Page 73 and 74: 1 H, 13 C NMR AND GIAO/DFT CALCULAT
- Page 75 and 76: NMR STUDY OF THE HUMIC ACIDS EXTRAC
- Page 77 and 78: NMR STUDY OF LAYERED ANGANITE La 1.
- Page 79 and 80: MAGNETIC SUSCEPTIBILITY EVALUATION
- Page 81 and 82: A NOVEL SOURCE OF MAGNETIC FIELD FO
- Page 83 and 84: DETERMINATION OF AMINO ACIDS IN BOD
- Page 85 and 86: MOLECULAR DYNAMICS OF TERT-BUTYL CH
- Page 87 and 88: MR MICROSCOPY IN COMPARISON OF YOUN
- Page 89 and 90: NH CH CH 2 NH CH CO COONa n CH 2 m
- Page 91 and 92: 35 Cl-NQR STUDY OF MOLECULAR DYNAMI
- Page 93 and 94: 35 Cl-NQR STUDY OF THE SUBSTITUENT
- Page 95 and 96: 13 C AND 1 H NUCLEAR MAGNETIC SHIEL
- Page 97 and 98: 13 C CPMAS NMR OF ANTHOCYANINS AND
- Page 99 and 100:
INVESTIGATION OF TRANSESTERIFICATIO
- Page 101 and 102:
T 2 RELAXATION MAP OF ARTICULAR CAR
- Page 103 and 104:
MICROTOMOGRAPHY STUDIES OF TABLETS
- Page 105 and 106:
MAPPING OF THE BI-EXPONENTIAL DIFFU
- Page 107 and 108:
DEUTERIUM NMR IN RMn 2 D 2 (R = Y,
- Page 109 and 110:
The neutron scattering (IINS, QNS,
- Page 111 and 112:
STRUCTURE OF 4-QUINOLINONES ANALYSE
- Page 113:
NMR MULTIPOLE RELAXATION IN THE ROT