Report No xxxx - Instytut Fizyki JÄ drowej PAN
Report No xxxx - Instytut Fizyki JÄ drowej PAN
Report No xxxx - Instytut Fizyki JÄ drowej PAN
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
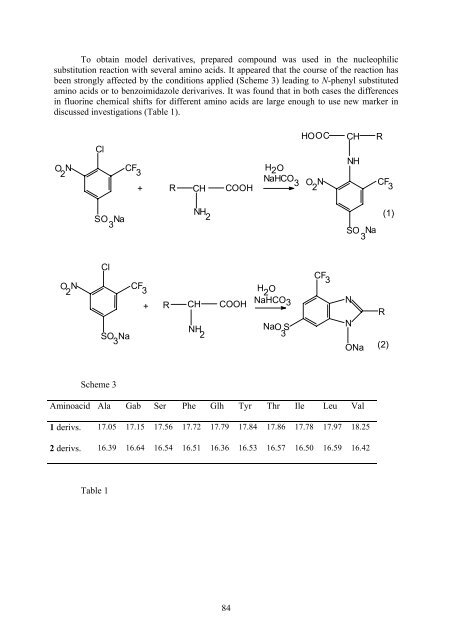
To obtain model derivatives, prepared compound was used in the nucleophilic<br />
substitution reaction with several amino acids. It appeared that the course of the reaction has<br />
been strongly affected by the conditions applied (Scheme 3) leading to N-phenyl substituted<br />
amino acids or to benzoimidazole derivarives. It was found that in both cases the differences<br />
in fluorine chemical shifts for different amino acids are large enough to use new marker in<br />
discussed investigations (Table 1).<br />
O N<br />
2<br />
Cl<br />
CF<br />
3<br />
+<br />
R<br />
CH<br />
COOH<br />
H 2 O<br />
NaHCO<br />
3<br />
HOOC<br />
O 2<br />
N<br />
CH<br />
NH<br />
R<br />
CF<br />
3<br />
SO 3<br />
Na<br />
NH<br />
2<br />
SO 3<br />
Na<br />
(1)<br />
Cl<br />
O N<br />
CF 2 3<br />
+ R CH COOH<br />
NH<br />
SO Na<br />
2<br />
3<br />
H 2<br />
O<br />
NaHCO 3<br />
NaO S<br />
3<br />
CF 3<br />
N<br />
N<br />
ONa<br />
R<br />
(2)<br />
Scheme 3<br />
Aminoacid Ala Gab Ser Phe Glh Tyr Thr Ile Leu Val<br />
1 derivs. 17.05 17.15 17.56 17.72 17.79 17.84 17.86 17.78 17.97 18.25<br />
2 derivs. 16.39 16.64 16.54 16.51 16.36 16.53 16.57 16.50 16.59 16.42<br />
Table 1<br />
84