H e m a t o lo g y E d u c a t io n - European Hematology Association
H e m a t o lo g y E d u c a t io n - European Hematology Association
H e m a t o lo g y E d u c a t io n - European Hematology Association
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
or higher. It is, however, important to understand that<br />
these regimens have not been prospectively studied in<br />
young adults; rather they have been adopted. Pediatric<br />
regimens are generally more intensive, include far more<br />
aspariginase, and reflect a greater protocol discipline,<br />
especially about timeliness. 73<br />
Philadelphia chromosome-positive<br />
Until recently, Philadelphia chromosome-positive<br />
ALL (Ph+) was considered the ALL with the poorest<br />
prognosis. With chemotherapy a<strong>lo</strong>ne, most patients did<br />
not survive one year. 74–79 Al<strong>lo</strong>geneic transplant improved<br />
the survival significantly 78,80–84 and became the gold standard<br />
for treatment. The advent of tyrosine kinase<br />
inhibitors (TKIs) has completely revolut<strong>io</strong>nized the<br />
treatment of Ph+ ALL and has become a part of a new<br />
gold standard.<br />
The most important issues regarding the treatment of<br />
Ph+ ALL in the TKI are:<br />
Induct<strong>io</strong>n<br />
• How to combine TKI with chemotherapy?<br />
• Which TKI to choose?<br />
• TKI and CNS<br />
Postinduct<strong>io</strong>n<br />
• In the TKI era, is al<strong>lo</strong>transplant still a must?<br />
Maintenance<br />
• TKI after transplant?<br />
Induct<strong>io</strong>n. The combinat<strong>io</strong>n of convent<strong>io</strong>nal chemo -<br />
therapy and imatinib mesylate has improved the CR<br />
rate and the OS compared to historical controls<br />
[reviewed in ref. #85)]. The optimal dose of imatinib is<br />
London, United Kingdom, June 9-12, 2011<br />
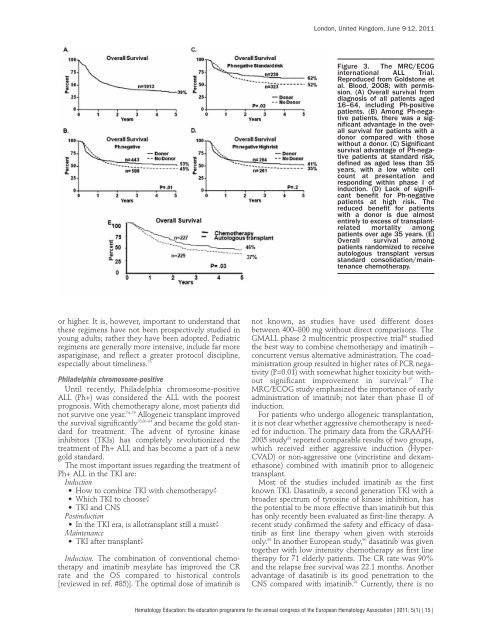
Figure 3. The MRC/ECOG<br />
internat<strong>io</strong>nal ALL Trial.<br />
Reproduced from Goldstone et<br />
al. B<strong>lo</strong>od, 2008; with permiss<strong>io</strong>n.<br />
(A) Overall survival from<br />
diagnosis of all patients aged<br />
16–64, including Ph-positive<br />
patients. (B) Among Ph-negative<br />
patients, there was a significant<br />
advantage in the overall<br />
survival for patients with a<br />
donor compared with those<br />
without a donor. (C) Significant<br />
survival advantage of Ph-negative<br />
patients at standard risk,<br />
defined as aged less than 35<br />
years, with a <strong>lo</strong>w white cell<br />
count at presentat<strong>io</strong>n and<br />
responding within phase I of<br />
induct<strong>io</strong>n. (D) Lack of significant<br />
benefit for Ph-negative<br />
patients at high risk. The<br />
reduced benefit for patients<br />
with a donor is due almost<br />
entirely to excess of transplantrelated<br />
mortality among<br />
patients over age 35 years. (E)<br />
Overall survival among<br />
patients randomized to receive<br />
auto<strong>lo</strong>gous transplant versus<br />
standard consolidat<strong>io</strong>n/maintenance<br />
chemotherapy.<br />
not known, as studies have used different doses<br />
between 400–800 mg without direct comparisons. The<br />
GMALL phase 2 multicentric prospective trial 86 studied<br />
the best way to combine chemotherapy and imatinib –<br />
concurrent versus alternative administrat<strong>io</strong>n. The coadministrat<strong>io</strong>n<br />
group resulted in higher rates of PCR negativity<br />
(P=0.01) with somewhat higher toxicity but without<br />
significant improvement in survival. 87 The<br />
MRC/ECOG study emphasized the importance of early<br />
administrat<strong>io</strong>n of imatinib; not later than phase II of<br />
induct<strong>io</strong>n.<br />
For patients who undergo al<strong>lo</strong>geneic transplantat<strong>io</strong>n,<br />
it is not clear whether aggressive chemotherapy is needed<br />
for induct<strong>io</strong>n. The primary data from the GRAAPH-<br />
2005 study 88 reported comparable results of two groups,<br />
which received either aggressive induct<strong>io</strong>n (Hyper-<br />
CVAD) or non-aggressive one (vincristine and dexamethasone)<br />
combined with imatinib pr<strong>io</strong>r to al<strong>lo</strong>geneic<br />
transplant.<br />
Most of the studies included imatinib as the first<br />
known TKI. Dasatinib, a second generat<strong>io</strong>n TKI with a<br />
broader spectrum of tyrosine of kinase inhibit<strong>io</strong>n, has<br />
the potential to be more effective than imatinib but this<br />
has only recently been evaluated as first-line therapy. A<br />
recent study confirmed the safety and efficacy of dasatinib<br />
as first line therapy when given with steroids<br />
only. 89 In another <strong>European</strong> study, 90 dasatinib was given<br />
together with <strong>lo</strong>w intensity chemotherapy as first line<br />
therapy for 71 elderly patients. The CR rate was 90%<br />
and the relapse free survival was 22.1 months. Another<br />
advantage of dasatinib is its good penetrat<strong>io</strong>n to the<br />
CNS compared with imatinib. 91 Currently, there is no<br />
Hemato<strong>lo</strong>gy Educat<strong>io</strong>n: the educat<strong>io</strong>n programme for the annual congress of the <strong>European</strong> Hemato<strong>lo</strong>gy Associat<strong>io</strong>n | 2011; 5(1) | 15 |