liiiMIIIfl~UDliiiMIII~U - Biblioteca de la Universidad Complutense ...
liiiMIIIfl~UDliiiMIII~U - Biblioteca de la Universidad Complutense ...
liiiMIIIfl~UDliiiMIII~U - Biblioteca de la Universidad Complutense ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
fliE JOURNAI. OF WOWG¡CM. CHEMISTRY <br />
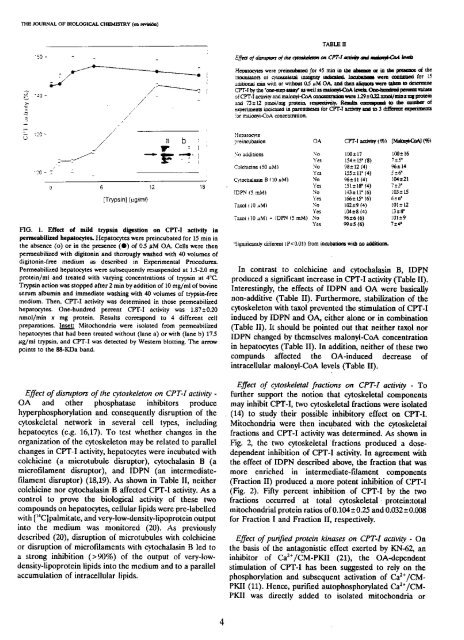
HG. 1. FIlen of n.ild brypsin digestion en CPT-I actidby <strong>la</strong><br />
penneabilized hepatocytes. Hopatocytos were preincutiated for 15 mm ira<br />
dio absonce (o) or ira Iba presoraco (@)of 0.5 gM OA. Ccli; ~cre ehen<br />
pormoabilized váth digitorain arad thorougJy washod with 40 volumes cf<br />
digitonin-free modium as <strong>de</strong>scribed ira Experimental Procedures.<br />
Permoabulized hopatocytes were subsequoratly resuspendod at 1.5-2.0 mg<br />
protejo/ml and troated with vatying concentrations of tzypsin at 4C.<br />
Trypsin action was stoppcd aftor 2 mira ti>’ additiora of 10 mg/ml of bovino<br />
serum albuniin arad immodiate washing with 40 volumos of erypsin-froe<br />
medium. mora. CPT-T activity was <strong>de</strong>tormined ira thoso penneabilizod<br />
hepatocytes. One-bundrod percoral CPT-i activity was 1.87±0.20<br />
ramol/min ir mg protoira. Resu¡ts corrospond te 4 d¡fferont cali<br />
preparation;. lnset: Mitochondria were iso<strong>la</strong>tod from pormeabulized<br />
hepatocytos that liad beco troatod wiLbout (<strong>la</strong>ne a) or váth (<strong>la</strong>rio ti) 17.5<br />
gg/mI tzypsin, arad CPT-I was <strong>de</strong>tected ti>’ Westorn blctting. ‘Dio arrow<br />
points te iba 88-KDa band.<br />
Effect of disn¿ptors of tite cytoskeleton oit CpT-i ac¡iviry -<br />
OA and otber phosphataso inhibitors produce<br />
hyperphosphory<strong>la</strong>rion and corasoquontly disruption of the<br />
cytoskeletal network ira sevaral cail types, including<br />
hopatocytes (e.g. 16,17). To test whether changas in Lho<br />
organization of the cytoske¡eton may be re<strong>la</strong>ted to parallel<br />
changas ira CPT-I activity, hepatocytcs were incubaLod wath<br />
colchicune (a microtubule disrupLor), cytocha<strong>la</strong>sin B (a<br />
m¡crofi<strong>la</strong>mont disrupror), and IDPN (ara intermediatefi<strong>la</strong>ment<br />
disruptor) (18,19). As shown in Table II, raeithor<br />
coichicine nor cytocha<strong>la</strong>sun B affected CPT-I acrivity. A; a<br />
control to prova the biologica] activity of Lhese Nro<br />
compounds on hepatocytes, cellu<strong>la</strong>r lipid; were pre-<strong>la</strong>beiled<br />
with [‘<br />
4C]palmitate, and very-low-<strong>de</strong>rasity-lipoprotein output<br />
anto the medium was monitorod (26). As previousiy<br />
<strong>de</strong>scribed (20), disruption of microtubules wirh colciiicine<br />
or disruption of microfi<strong>la</strong>monts with cytocha<strong>la</strong>sira 8 led to<br />
a sLrong inhibition (>90%) of the output of very-lowdonsity-lipoprotoin<br />
lipids into the medium arad to a paral<strong>la</strong>l<br />
accumu<strong>la</strong>Lion of intracellu<strong>la</strong>r lipids.<br />
»<br />
TABLE II<br />
Effces ofdin,,xas of¡he qeatkMon a. CPUn* .~ i0m4 frtei<br />
¡<br />
Hentccvtes were premoibetcd for 45 mira lo tite k — <strong>la</strong> ¡be ptn of tite<br />
nnowaaon of cvnkcleuá inujmny indialsd. ¡aaÉs~ we,t anaS ter 15<br />
adadonal — váab or “.,thout 0.5 »MOA. aaá din .Iiquoc .exe mkn te <strong>de</strong><strong>la</strong>nune<br />
cfl”-i by tite one-etepsafas w.I¡ ma wuloa>4.CoA¡o.k Cw.4n~S pan van<br />
of CFI-! actnwv andmalouiyl-CaAaeennnozwere ).29±ftflo,neI/mnnz mgpmtelfl<br />
and 73±12pmoi/mg protein. respeaivciy. Rh. an~k