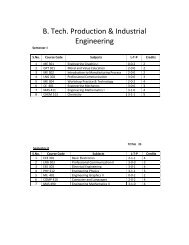
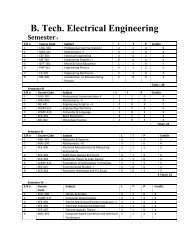
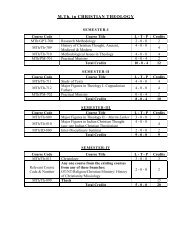
Study of Hygienic practices of street food vendors in ... - Shiats.edu.in
Study of Hygienic practices of street food vendors in ... - Shiats.edu.in
Study of Hygienic practices of street food vendors in ... - Shiats.edu.in
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Aradhana Irene Charan, Nishant Kumar Srivastava, Sanjay S<strong>in</strong>gh, Amit Alexander Charanand Abhishek Sharanacids, mak<strong>in</strong>g it a richer source <strong>of</strong> prote<strong>in</strong>s compared to other vegetables. The drumsticktree <strong>in</strong> fact has the highest prote<strong>in</strong> ratio <strong>of</strong> any plant on earth, with calcium content <strong>of</strong> theleaves be<strong>in</strong>g 297 mg per 100 g <strong>of</strong> leaves. 100 g <strong>of</strong> freshly cooked drumstick leavesprovide for 75 % <strong>of</strong> iron and 50 % <strong>of</strong> prote<strong>in</strong>s required by a grow<strong>in</strong>g child.MATERIALS AND METHODFresh leaves <strong>of</strong> M. oleifera were collected from Germplasm Bank established atNational Bureau <strong>of</strong> Plant Genetic Resources (NBPGR), Ranchi, which conta<strong>in</strong>s accession<strong>of</strong> drumstick plants which were collected from different locations <strong>of</strong> Jharkhand. Extraction<strong>of</strong> DNA <strong>in</strong> M. oleifera was done by (Doyle and Doyle, 1990), with some modification<strong>in</strong> the proc<strong>edu</strong>re, as M. oleifera conta<strong>in</strong>s some phenolic compounds which causestick<strong>in</strong>ess <strong>in</strong> the sample and make it difficult to isolate the DNA . So, <strong>in</strong> context withabove an additional step is added, which uses wash buffer conta<strong>in</strong><strong>in</strong>g 0.1% PVP and4% - Mercaptoethanol to wash out the phenolic compounds (Hedrick, 1992).The genomic DNA was then subjected to spectrophotometric quantification at 260nm and 280 nm. 14 eppendorf tubes were taken each hav<strong>in</strong>g 1 ml volume consist<strong>in</strong>g <strong>of</strong>980 µl TE buffer and 20 µl DNA sample and another 1 ml eppendorf tube which is usedas blank consist<strong>in</strong>g <strong>of</strong> 1000 µl TE buffer. Agarose gel electrophoresis was also done tocheck the presence <strong>of</strong> DNA <strong>in</strong> the sample. RAPD analysis was done with total sevenprimers which were already available <strong>in</strong> the laboratory and they were selected on thebasis <strong>of</strong> their genetic diversity and identity (Table 1). PCR was performed with follow<strong>in</strong>gsett<strong>in</strong>gs- total 18 µl <strong>in</strong> which 1.5 µl dNTPs, 0.5 µl Taq Polymerase, 1.0 µl Taq Buffer, 1.5µl Primer, 0.5 µl MgCl 2, 11 µl PCR grade water and 2 µl <strong>of</strong> DNA sample. Total 45 cycleswere performed. Amplification products were analyzed by electrophoresis us<strong>in</strong>g 1.5%Agarose gel <strong>in</strong> 1X TAE runn<strong>in</strong>g buffer. The bands were visualized under the GelDocumentation System. Bands were scored, and analyzed with s<strong>of</strong>tware POPGENEVersion 1.32 (32-bit).RESULTS AND DISCUSSIONIn prelim<strong>in</strong>ary screen<strong>in</strong>g, seven primers were chosen for the evaluation <strong>of</strong> geneticdiversity among the M. oleifera populations. Specific DNA band patterns were observedwith all the seven primers used. A total <strong>of</strong> twenty five bands were scored and all werepolymorphic (Arnold and Emms, 1998). On average each primer produced 3.2 RAPDbands, the largest number was obta<strong>in</strong>ed from RPI 8 and RPI 15 and lowest number wasobserved from RPI 5 after runn<strong>in</strong>g on Agarose gel. UPGMA cluster was prepared us<strong>in</strong>gNei's Genetic distance method UPGMA (Nei, 1973) which is modified from NEIGHBOR107