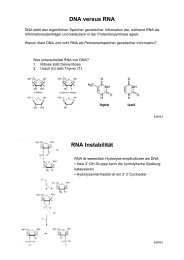
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
poral requirement for trx in or<strong>der</strong> to achieve normal<br />
expression patterns. We have shown with X-CHIP a<br />
dynamic association of TRX with CMMs and promoters<br />
of the BX-C and found a close cooperation of TRX<br />
with PcG members at CMMs.<br />
Several members of the Drosophila trxG have counterparts<br />
in humans and mice. The mixed-lineage leukaemia<br />
(MLL) gene (also known as ALL-1, HRX), a<br />
TRX homologue, was found through its implication in<br />
the majority of infantile acute lymphocytic and mixed<br />
lineage leukemias. Disruption of the murine Mll gene<br />
by gene targeting causes homeotic transformations of<br />
the vertebrae in heterozygotes and loss of homeotic<br />
gene expression, supporting the notion that Mll is a<br />
functional equivalent of trx.<br />
To date, not much is known at the molecular level<br />
how translocation of MLL with various other chromosomal<br />
locations generate leukemia. At least 23 fusion<br />
MLL proteins involved in leukemia have been identified.<br />
As part of our collaborative efforts with<br />
the Canaani lab, we have generated transgenic flies<br />
expressing full length MLL, and fusion proteins<br />
MLL-AF9, MLL-AF4, and MLL-AF17 un<strong>der</strong> GAL4<br />
control. Expression of these proteins in Drosophila<br />
may reveal their effects by determining with which<br />
pathway in Drosophila development they influence.<br />
Crosses between our transgenic flies and various<br />
GAL4 drivers revealed that only MLL-AF9 and<br />
MLL-AF17 constructs had a slight effect on Drosophila<br />
development. When we took a closer look at the<br />
molecular level, we could not detect the proteins on<br />
Western, but at the RNA level, we see transcripts from<br />
all four constructs. Interestingly, this same effect is<br />
also observed with MLL-AF9 in mouse. We are currently<br />
testing whether overexpression of the murine<br />
proteins results in cell death as recent published observations<br />
link MLL with apoptosis.<br />
100<br />
IV. Drosophila as a model system to identify<br />
components involved in the processing<br />
and the function of the human Amyloid Precursor<br />
Protein<br />
G. Merdes, B. Brückner, DFG (in collaboration<br />
with K. Beyreuther (<strong>ZMBH</strong>))<br />
The human amyloid precursor protein (APP) is an<br />
integral membrane protein with two homologues in<br />
invertebrates: the amyloid protein-like protein 1 (C.<br />
elegans, APL-1) and the amyloid precursor proteinlike<br />
protein (D. melanogaster, APPL). Flies deficient<br />
for expression of APPL show a phototaxis impairment<br />
that can be rescued by the expression of human APP<br />
suggesting an evolutionary conservation of APP function.<br />
The importance of APP in the pathogenesis of<br />
Alzheimer’s disease (AD) became apparent through<br />
the identification of distinct mutations in the gene<br />
causing early onset familial AD. The major component<br />
of the plaques and amyloid found in senile Alzheimer<br />
patients is a proteolytic product of APP, the<br />
βA4 peptide. APP is cleaved in vivo by several proteases<br />
termed α−, β−, and γ−secretase. Cleavage of<br />
APP by the α−secretase prevents the production of<br />
βA4. Despite the availability of APP-null mutants and<br />
transgenic mice expressing human APP, the physiological<br />
role of APP remains unknown and many factors<br />
involved in the processing of APP have not yet<br />
been clearly identified. Therefore we have established<br />
Drosophila melanogaster as a model system to study<br />
the function and processing of APP.<br />
Various forms of human APP were expressed in<br />
Drosophila tissue culture cells as well as in transgenic<br />
fly lines. Studies in both systems revealed that also in<br />
flies APP is processed in a similar way as in mammalian<br />
cells in regard to the α- and γ-cleavages. In addition,<br />
transgenic flies expressing full-length forms of<br />
APP in the wing imaginal discs are characterized by a<br />
blistered-wing phenotype, suggesting that the expression<br />
of human APP interferes with the cell adhesion<br />
between the dorsal and ventral epithelial cell monolayers<br />
of the wing (Figure 3). To define the involvement<br />
of APP in the blistered wing phenotype in more<br />
detail as well as to identify factors involved in the processing<br />
of APP, we perform biochemical and genetic<br />
studies. Since the observed wing phenotype depends<br />
on full-length APP, an increased processing of APP,<br />
e.g. by the simultaneous overexpression of an APPspecific<br />
protease, results in a suppression of the phenotype.<br />
This observation enables us to identify proteins<br />
involved in the processing of APP by the use of<br />
genetic screens and to subsequently identify the corresponding<br />
mammalian homologues. In a similar way,<br />
gene products required for the effect of APP on cell<br />
adhesion and therefore for APP function, can also be<br />
identified by genetic screens. Mutations in these genes<br />
result in suppression or an enhancement of the blis-<br />
tered wing phenotype, dependent on their function.<br />
Drosophila can be mutagenized by the use of chemicals<br />
or by the use of the transposable P-element.<br />
The advantage of P-element mutagenesis is the easier<br />
identification of the mutagenized genes by isolating<br />
and sequencing the flanking genomic DNA. Therefore<br />
we have recently performed a P-element based genetic<br />
screen as well as screened collections of already existing<br />
Drosophila mutants. The analysis of 150 000 flies<br />
with potential new P-element insertion sites resulted<br />
in the isolation of several enhancer- and suppressormutations,<br />
together with several genes interfering with<br />
wing patterning. The corresponding genes, which we<br />
are currently characterizing further, encode factors<br />
involved in signal transduction, maintenance of cell<br />
shape and cell adhesion, and protein processing.<br />
We utilize the fruit fly Drosophila melanogaster as<br />
a genetic model system. Since Thomas Hunt Morgan<br />
introduced Drosophila as a model organism into genetics<br />
at the beginning of this century and isolated the<br />
Figure 3. Expression of different<br />
isoforms of human APP in transgenic<br />
Drosophila. Only full-length<br />
APP result in a blistered wing<br />
phenotype.<br />
101