ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
APP catalyzed the reduction of H 2 O 2 and oxidation of<br />
Cu(I) to Cu(II) in a „peroxidative“ reaction in vitro.<br />
The resulting bound copper-hydroxyl radical intermediate<br />
[APP-Cu(II) (•OH)] then likely participated in<br />
a Fenton type of reaction with radical formation as a<br />
prerequisite for APP-Cu(I) complex degradation. Evidence<br />
from two observations suggests that the reaction<br />
takes place in two phases. Bathocuproine, a trapping<br />
agent for Cu(I), abolished the initial fragmentation<br />
of APP, and chelation of Cu(II) by DTPA (diethylenetriaminepentaacetic<br />
acid) interrupted the reaction<br />
cascade induced by H 2 O 2 at later stages. WT cortical,<br />
cerebellar and hippocampal neurons were significantly<br />
more susceptible than their respective APP 0/0 neurons<br />
to toxicity induced by physiological concentrations<br />
of copper but not by zinc or iron. There was no difference<br />
in copper toxicity between APLP2 0/0 and WT<br />
neurons, demonstrating specificity for APP-associated<br />
copper toxicity. Treatment of neuronal cultures with a<br />
peptide corresponding to the human APP copper binding<br />
domain (APP142-166) potentiated copper but not<br />
iron or zinc toxicity. Incubation of APP142-166 with<br />
low density lipoprotein (LDL) and copper resulted<br />
in significantly increased lipid peroxidation compared<br />
to copper and LDL alone. Substitution of the<br />
copper co-ordinating histidine residues with asparagines<br />
(APP142-166 H147N, H149N, H151N ) abrogated the<br />
toxic effects. A peptide corresponding to the zinc binding<br />
domain (APP181-208) failed to induce copper or<br />
zinc toxicity in neuronal cultures. These data support a<br />
role for the APP copper binding domain in APP-mediated<br />
Cu(I) generation and toxicity in primary neurons.<br />
To test the hypothesis that biometals such as copper<br />
can contribute to the amyloid pathology in AD we are<br />
currently investigating the dietary exposure of copper<br />
and its chelators to APP transgenic mice and to APP<br />
42<br />
knock-out mice. The effects of dietary supplementation<br />
is studied by ICP-MS (inductively coupled plasma<br />
mass-spectrometry) to determine the levels of Cu, Zn<br />
and Fe. The AD pathology is analyzed by conventional<br />
methods. We expect that Cu has an effect on the<br />
AD pathology and metal-chelation can delay amyloid<br />
Aß deposition in APP transgenic mice.<br />
II. The role of copper and zinc in the normal<br />
function of the amyloid precursor protein<br />
(APP)<br />
C. Schmidt, A. Simons, M. Strauss, A. Schlicksupp,<br />
G. Multhaup (in collaboration with D.<br />
Ha-Hao, R. Cappai, A. White, C.L. Masters and<br />
K. Beyreuther)<br />
The expression of APP and APLP2 in the brain<br />
suggests they could have an important direct or indirect<br />
role in neuronal metal homeostasis. In APP and<br />
APLP2 knockout mice copper levels were significantly<br />
elevated in both APP 0/0 and APLP2 0/0 cerebral<br />
cortex (40% and 16%, respectively) and liver (80% and<br />
36%, respectively) compared with matched wild-type<br />
(WT) mice. These findings indicate APP and APLP2<br />
expression specifically modulates copper homeostasis<br />
in the liver and cerebral cortex, the latter being a<br />
region of the brain particularly involved in AD. Perturbations<br />
to APP metabolism and in particular, its secretion<br />
or release from neurons may alter copper homeostasis<br />
and explain a disturbed metal-ion homeostasis<br />
observed in AD.<br />
Zinc up to concentrations of 50µM or the presence of<br />
1,10-phenanthroline specifically increased the level of<br />
secreted APP in APP transfected CHO-K1 cells. By<br />
contrast, the level of secreted APP in copper-resistant<br />
CHO-CUR3 cells remained unaffected. APP holoprotein<br />
increased dramatically in CHO-CUR3 cells com-<br />
pared with CHO-K1 cells. The large decrease of Aß<br />
release seen in both cell lines at elevated extracellular<br />
zinc levels was due to specific inhibition of secretion.<br />
These results indicate that a disturbed zinc-homeostasis<br />
may be an important factor influencing APP production,<br />
transport and processing.<br />
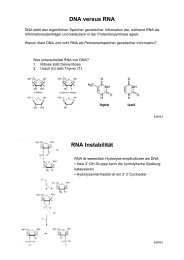
Figure 3: The α- and β-secretase protease activities cleave<br />
APP within its ectodomain. The remaining membranebound<br />
C-terminal fragment p3CT (after α-secretase cleavage<br />
of APP) is further cleaved by γ-secretase in the middle<br />
of the putative transmembrane domain, yielding p3 (α- and<br />
γ-secretase activities). Amyloid Aβ is produced by β- and<br />
γ-secretase activities; CuBD-I, copper binding domain 1.<br />
Adding copper to APP-transfected CHO cells greatly<br />
reduced the levels of ß-amyloid (Aß) peptide in both<br />
parental CHO-K1 and in copper resistant CHO-CUR3<br />
cells which have lower intracellular copper levels.<br />
Copper also caused an increase in the secretion of the<br />
APP ectodomain indicating that the large decrease in<br />
Aß release was not due to a general inhibition in protein<br />
secretion. There was an increase in intracellular<br />
full-length APP levels which paralleled the decrease<br />
in Aß generation suggesting the existence of two distinct<br />
regulating mechanisms, one acting on Aß production<br />
and the other on APP synthesis. Thus, our<br />
findings suggest that copper or copper agonists might<br />
be useful tools to discover novel targets for anti-<br />
Alzheimer drugs since copper promoted the non-amyloidogenic<br />
pathway of APP.<br />
Our current un<strong>der</strong>standing is that copper and/or zinc<br />
binding is central to the normal cellular function of<br />
APP. We therefore want to identify agonists of APP<br />
ligand binding sites (e.g. of the copper-binding site)<br />
that are able to inhibit amyloidogenic proteolytic processing<br />
of APP. Lead compounds will be discovered<br />
followed by a further screen for small APP ligands.<br />
Our research program combines biochemical, immunohistochemical<br />
and cellular approaches to start a<br />
risk-benefit analysis of divalent metal binding agonists<br />
of APP in vitro.<br />
III. Dimerization and stability of APP isoforms:<br />
influence of APP ligands on relative<br />
stability and metabolism<br />
M. Strauss, C. Schmidt, A. Simons, A. Schlicksupp,<br />
C. Elle, G. Multhaup (in collaboration with<br />
C. Haass, C.L. Masters and K. Beyreuther)<br />
The highly conserved nature and tissue specificity of<br />
the eight APP isoforms provide circumstantial evidence<br />
that functional differences among isoforms may<br />
exist in vitro and in vivo. When APP and Aß are central<br />
to AD pathogenesis, then molecules which influence<br />
the conformation and the stability of APP isoforms<br />
and that interact with specific APP forms could<br />
differentially alter their metabolism/activity and thus<br />
represent risk factors for AD.<br />
For example, heparin and Zn(II) were found to augment<br />
the ability of full-length and secreted KPI-APP to<br />
inhibit FXIa. In contrast, both compounds heparin and<br />
43