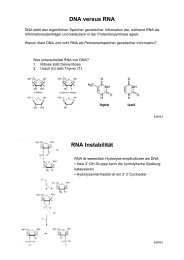
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
telt intrazelluläre Signaltransduktion.<br />
Klöcker, Uta (<strong>2000</strong>): Regulation <strong>der</strong> hepadnaviralen<br />
Replikation: Untersuchung des Effektes antiviraler<br />
Mediatoren mit Hilfe hepadnaviraler Vektoren.<br />
Sprinzl, Martin Franz (<strong>2000</strong>): Der adenovirale Genomtransfer<br />
überwindet die Speziesbarriere <strong>der</strong> Hepatitis-<br />
B-Viren.<br />
Habilitations<br />
Hug, Hubert (1998). Signalübertragung <strong>der</strong> CD95<br />
(Fas/APO-1)-vermittelten Apoptose.<br />
Knolle, Percy (<strong>2000</strong>). Die Leber als immunregulatorisches<br />
Organ: Bedeutung <strong>der</strong> sinusoidalen Endothelzellen<br />
und Kupffer Zellen.<br />
Protzer, Ulrike (<strong>2000</strong>). Virus-Wirt-Interaktion bei <strong>der</strong><br />
Hepatitis B Virus Infektion: vom verbesserten Verständnis<br />
zur Entwicklung neuer Therapien.<br />
Urban, Stephan (<strong>2000</strong>). Identifizierung des aviären<br />
Hepatitis B Virus Rezeptors und Charakterisierung<br />
seiner Interaktion mit dem viralen Hüllprotein.<br />
STRUCTURE OF THE GROUP<br />
E-mail: hshd@zmbh.uni-heidelberg.de<br />
Group lea<strong>der</strong> Schaller, Heinz, Prof. Dr.<br />
Research associates/<br />
postdoctoral<br />
fellows Knaust, Andreas, Dr. *<br />
Kuhn, Christa, Dr.<br />
Mabit, Hélène, Dr.*<br />
124<br />
Obert, Sabine, Dr.*<br />
Protzer, Ulrike, Dr.<br />
Sirma, Hüseyin, Dr.*<br />
Urban, Stephan, Dr.<br />
Visiting<br />
scientists Wimmer, Eckard, Prof.*<br />
from SUNY at Stony Brook, USA<br />
(Humboldt Awardee)<br />
Grgacic, Elizabeth, Dr.*<br />
from Macfarlane Burnet Centre,<br />
Fairfield, Australia<br />
Ph.D. students Breiner, Klaus, Dipl.Ing.,<br />
MA chem.<br />
Dumortier, Jêrome*<br />
Klöcker, Uta<br />
Rothmann, Kirsten*<br />
Sprinzl, Martin*<br />
Thake, Sandra*<br />
Techn.<br />
assistants Glass, Bärbel, Dipl. Biol.<br />
Götzmann, Martina*<br />
Oberwinkler, Heike*<br />
Zachmann-Brand, Beate<br />
*) part of the time reported<br />
Project Group Percy Knolle<br />
Regulation of the Immune Response in the<br />
Liver<br />
The liver is a unique organ with regard to the induction<br />
of peripheral immune tolerance. Delivery of antigen<br />
into the liver results in tolerance induction towards this<br />
specific antigen as is seen by the increased survival<br />
of organ transplants following portal venous injection<br />
of donor antigens or following portal venous drainage<br />
of the organ transplant. A number of different hepatic<br />
cell populations and the unique hepatic microenvironment<br />
seem to cooperate to induce immune tolerance.<br />
We are studying the contribution to immune tolerance<br />
of the sinusoidal endothelial cells of the liver (LSEC),<br />
which interact constitutively with leukocytes in the<br />
sinusoidal lumen (see Fig. 1) as well as two other<br />
Figure 1. Schematic drawing of the relative position and<br />
cell sizes in the hepatic sinusoid. Leukocytes are in intimate<br />
contact with LSEC during liver passage and constitutively<br />
adhere to LSEC un<strong>der</strong> physiologic conditions.<br />
hepatic sinusoidal cell populations, the Kupffer cells<br />
and the liver associated lymphocytes. Kupffer cells<br />
and LSEC have the capacity to act as antigen-presenting<br />
cells and activate CD4 + T cells suggesting<br />
immune-surveillance function for the liver. However,<br />
antigen-presentation by LSEC and Kupffer cells is<br />
stringently controlled by physiological constituents<br />
of portal venous blood (e.g. endotoxin) and by both,<br />
autocrine and paracrine action of soluble mediators,<br />
which are induced in resident liver cells by endotoxin.<br />
Activation of naive CD4 + T cells by LSEC outside<br />
lymphoid tissue may constitute one mechanism of<br />
hepatic tolerance induction, because antigen-presention<br />
by LSEC leads to the generation of CD4 + T cells<br />
that express immunosuppressive cytokines and act as<br />
regulatory T cells.<br />
I. Functional inactivation of CD8+ T cells<br />
by cross-presenting sinusoidal endothelial<br />
cells<br />
J. Ohl und A. Limmer<br />
Presentation of endocytosed antigens on MHC-class<br />
I molecules on antigen-presenting cells to cytotoxic<br />
CD8 + T cells (cross-presentation) is important for<br />
immunity to intracellular pathogens and was supposed<br />
to be restricted to dendritic cells and macrophages.<br />
Once in the blood, antigen is taken up efficiently by<br />
LSEC, but not significantly by other cell populations<br />
of the liver (Fig. 2). In vitro analysis of isolated<br />
LSEC reveiled that cross-presentation of endocytosed<br />
antigen to a antigen-specific CD8 + T cell hybridoma<br />
occured in LSEC. Cross-presentation in LSEC compared<br />
to other antigen-presenting cells requires only<br />
125