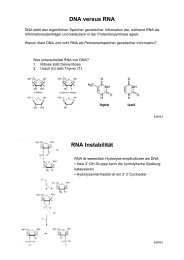
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
containing a subset of mRNAs are transported to<br />
specific regions inside the cell. This mechanism of<br />
local translation of transported mRNAs might play<br />
an important role to support protein targeting. The<br />
mRNA-binding protein vigilin displays unique features<br />
making it a good candidate to be a player in this<br />
process. Vigilin cofractionates with free and membrane<br />
bound ribosomes and shows an intracellular distribution<br />
similar as the endoplasmatic reticulum (ER).<br />
The protein consists almost exclusivly of RNA-binding<br />
domains. These domains provide multiple binding<br />
sites for messenger and ribosomal RNAs. To study the<br />
function of this unique RNA-binding protein we use<br />
budding yeast as a model system. Yeast expresses the<br />
structurally closely related protein Scp160p, which<br />
shows a similar intracellular distribution as vigilin.<br />
The SCP160 gene is not required for cell growth but<br />
a mutant without SCP160 fail to localize one specific<br />
mRNA. The majority of Scp160 protein associates<br />
with translating ribosomes, suggesting a function<br />
in the control of translation. Strong expression of<br />
Scp160p abolishes growth and reduces translation<br />
rates of a subset of so far unknown proteins. Cytoplasmic<br />
steady state localisation of Scp160p is independent<br />
of bulk mRNA export from the nucleus supporting<br />
the idea that Scp160p function is primary cytoplasmic<br />
and that the protein is not exported from the<br />
nucleus as constituent of a mRNA complex. Scp160p<br />
bears a masked nuclear import signal indicating that<br />
the function of Scp160p might be regulated by relocation<br />
of the protein to the nucleus.<br />
Microsomal ATP-Transport in yeast<br />
G. Konrad, T. Schlecker and P. Mayinger in collaboration<br />
with V. Bankaitis, Birmingham, Alabama<br />
ATP Transport into the ER is essential for ATP con-<br />
64<br />
suming reactions inside the ER lumen, including protein<br />
translocation and protein folding. We have characterized<br />
this nucleotide transport reaction as a specific<br />
ADP/ATP antiport and we have identified the<br />
Sac1 protein as an important regulator for this process.<br />
Sac1p is an ER and Golgi membrane protein with<br />
homology to a number of yeast and mammalian inositol<br />
phosphatases. It was shown recently that the N-terminal<br />
portion of Sac1p displays polyphosphoinositide<br />
phosphatase activity with unique enzymatic specificity.<br />
Consistent with these findings we obtained strong<br />
evidence in vitro that microsomal ATP transport is<br />
regulated via specific phosphoinositides.<br />
Cellular role of Sac1p in phosphoinositide<br />
signaling<br />
A. Then<br />
Sac1p plays additional roles in the regulation of<br />
intracellular membrane trafficking, phospholipid metabolism<br />
and the actin cytoskeleton. To elucidate the cellular<br />
function of Sac1p in more detail, we performed<br />
a synthetic lethal screen. In this screen we identified a<br />
novel allele of the SLT2 gene, which results in a severe<br />
growth defect when combined with a disruption of the<br />
SAC1 gene. The SLT2 gene codes for a PKC1-dependent<br />
MAP kinase, which plays an important role in<br />
cell wall integrity and in organization of cortical actin<br />
and performs an essential function in the context of<br />
a sac1D background. This genetic interaction with<br />
SLT2 defines a novel important function of the SAC1<br />
gene in regulating cell wall biogenesis and polarized<br />
secretion. Epistatic analysis places the SAC1 gene parallel<br />
to the PKC1 dependent SLT2 MAP kinase, linking<br />
this kinase cascade to phosphoinositide signaling.<br />
External Funding<br />
During the period reported our research was supported<br />
by grants from the Deutsche Forschungsgemeinschaft<br />
(SFB 352 “<strong>Molekulare</strong> Mechanismen intrazellulärer<br />
Transportprozesse”, Graduiertenkolleg “<strong>Molekulare</strong><br />
Zellbiologie”), from the Landesforschungsschwerpunkt<br />
“Protein-Faltung und -Transport: Mechanismen<br />
und Pathobiochemie”, from the EU (“Signal Recognition<br />
Particle-Network”) and from the Fonds <strong>der</strong> Chemischen<br />
Industrie.<br />
PUBLICATIONS<br />
Martoglio, B., Hauser, S. and Dobberstein, B. (1998).<br />
Cotranslational translocation of proteins into microsomes<br />
<strong>der</strong>ived from the rough endoplasmic reticulum<br />
of mammalian cells. In Cell Biology, A Laboratory<br />
Handbook. Academic Press, 2nd ed. Vol. 2, pp.<br />
265-273.<br />
Graf, R., Brunner, J., Dobberstein, B. and Martoglio,<br />
B. (1998). Probing the molecular environment of proteins<br />
by site-specific photocrosslinking. In Cell Biology,<br />
A Laboratory Handbook. Academic Press 2nd ed.<br />
Vol 4, pp. 495-501.<br />
Martoglio, B. & Dobberstein, B. (1998). Signal<br />
sequences – more than just greasy peptides. Trends in<br />
Cell Biology 8, 410-415.<br />
Dube, P., Bacher, G., Stark, H., Müller, F., Zemlin, F.,<br />
van Heel, M. and Brimacombe, R. (1998). Correlation<br />
of the expansion segments in mammalian rRNA<br />
with the fine structure of the 80S ribosome; a cryoelectron<br />
microscopic reconstruction of the rabbit reticulocyte<br />
ribosome at 21A resolution. J. Mol. Biol. 279,<br />
403-421.<br />
Otter-Nilsson, M., Hendriks, R., Pecheur-Huet, E.I.,<br />
Hoekstra, D. and Nilsson, T. (1999). Cytosolic<br />
ATPases, p97 and NSF, are sufficient to mediate rapid<br />
membrane fusion. EMBO J. 18, 2074-2083.<br />
Gruss, O.J., Feick, P., Frank, R. and Dobberstein, B.<br />
(1999). Phosphorylation of components of the ER<br />
translocation site. Eur. J. Biochem. 260, 785-793.<br />
Schrö<strong>der</strong>, K., Martoglio, B., Hofmann, M., Hölscher,<br />
C., Hartmann, E., Prehn, S., Rapoport, T.A. and Dobberstein,<br />
B. (1999). Control of glycosylation of MHC<br />
class II-associated invariant chain by translocon-associated<br />
RAMP4. EMBO J. 18, 4804-4815.<br />
Bacher, G., Pool, M. and Dobberstein, B. (1999). The<br />
ribosome regulates the GTPase of the β-subunit of the<br />
signal recognition particle receptor. J. Cell Biol. 146,<br />
723-730.<br />
Wang, L. and Dobberstein, B. (1999). Oligomeric<br />
complexes involved in translocation of proteins across<br />
the membrane of the endoplasmic reticulum. FEBS<br />
Lett. 457, 316-22.<br />
Hofmann , M.W., Höning, S., Rodinov, D., Dobberstein,<br />
B., von Figura, K. and Bakke, O. (1999).<br />
The leucine-based sorting motifs in the cytoplasmic<br />
domain of the invariant chain are recognized by the<br />
medium chains of the clathrin adaptors AP1 and AP2.<br />
J. Biol. Chem. 274, 36153-36158.<br />
Kochendörfer, K.-U., Then, A. R., Kearns, B. G.,<br />
Bankaitis, V. A., and Mayinger, P. (1999). Sac1p plays<br />
a crucial role in microsomal ATP transport, which<br />
is distinct from its function in Golgi phospholipid<br />
metabolism. EMBO J. 18, 1506-1515.<br />
65