ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
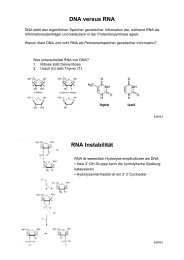
of individual proteins. The combination of peptide<br />
mass finger-printing and peptide fragmentation which<br />
matches the masses of in gel proteolytically generated<br />
peptides against theoretically digested proteins from<br />
the M. pneumoniae database, has proved to be very<br />
effective and reliable. Proteome analysis is limited by<br />
the sensitivity of mass spectrometry and the pH range<br />
of the first dimension of the two-dimensional gel electrophoresis.<br />
Presently proteins with isoelectric points<br />
Figure 1: 2-D-gel of a total protein extract from M. pneumoniae.<br />
First dimension (1-D) immobilized pH gradient,<br />
second dimension (2-D) 12,5% SDS-polyacrylamide gel.<br />
All stained protein spots (≈ 200) were assigned to the corresponding<br />
genes, but only the strongest spots are marked<br />
with a gene number.<br />
76<br />
Figure 2: Mock 2-D-gel of all proposed proteins of M.<br />
pneumoniae. Based on the predicted molecular mass and<br />
isoelectric point, each protein could be localized in this<br />
mock 2-D-gel. The yellow and blue dots represent the sum<br />
of all proposed proteins; the „blue“ proteins have been<br />
assigned to a gene.<br />
between 3-10 can only be separated routinely by<br />
immobilized pH gradients (Fig. 1), but the annotation<br />
of the genome sequence of M. pneumoniae predicts<br />
236 proteins with pIs greater than 10 (Fig. 2). When<br />
we analyze the protein extract of entire M. pneumoniae<br />
cells by two-dimensional gel electrophoresis<br />
and visualize the proteins by silver staining, about 450<br />
proteins can be detected and about 200 proteins by<br />
staining with the less sensitive Coomassie blue (Fig.<br />
1). So far, 200 proteins have been identified by mass<br />
spectrometry and assigned to the corresponding genes.<br />
We found in most cases a good correlation between<br />
DNA sequence based prediction of pI and mass of a<br />
protein and its final position within a two-dimensional<br />
gel. However, exceptions have been found where the<br />
experimentally <strong>der</strong>ived values were different. We suggest<br />
that in many of these exceptions posttranslational<br />
modification might have occurred. Furthermore,<br />
a comparison of transcriptome and proteome analysis<br />
shows that a relatively high copy number of a given<br />
transcript does not always correlate with a strong pro-<br />
tein signal and vice versa. Those genes could be interesting<br />
examples for regulation of gene expression at<br />
different levels. The proteome analysis is being done<br />
in cooperation with R. Frank, Heidelberg and A. Görg,<br />
München.<br />
IV. Is there a cytoskeleton-like structure in M.<br />
pneumoniae?<br />
J. Regula, A. Boonmee, W. Schaller<br />
The M. pneumoniae genome does not encode for a<br />
single gene known to be involved in the synthesis<br />
of a bacterial cell wall. M. pneumoniae is only surrounded<br />
by a cytoplasmic membrane that, exceptionally<br />
among bacteria, contains cholesterol as an essential<br />
component. Over the past twenty years evidence<br />
has accumulated that M. pneumoniae possesses a cytoskeleton-like<br />
structure, probably as a substitute for the<br />
missing cell wall. In analogy to the cytoskeleton of<br />
eukaryotic cells, such a structure could provide the<br />
necessary framework for maintaining and stabilizing<br />
the shape of M. pneumoniae, for motility and for the<br />
formation of an asymmetric cell.<br />
The first experimental evidence for a cytoskeleton-like<br />
structure in M. pneumoniae was provided by Meng<br />
and Pfister (1980) who detected fibrous structures<br />
by electron microscopy after treating M. pneumoniae<br />
cells with the nonionic detergent Triton X-100, which<br />
removed the membrane and the cytoplasm. These<br />
observations were confirmed and extended by several<br />
other researchers. These experiments and studies on<br />
the architecture and composition of eucaryotic cytoskeletons<br />
from cells which have been treated with the<br />
detergent Triton X-100 suggested that a cytoskeletonlike<br />
structure would also be enriched in the Triton<br />
X-100 insoluble fraction of M. pneumoniae.<br />
Therefore, we decided to determine the protein com-<br />
position of the Triton X-100 insoluble fraction of<br />
M. pneumoniae by 2-D-gel electrophoresis and mass<br />
spectrometry.<br />
Silver staining of 2-D gels of the Triton X-100 insoluble<br />
fraction revealed about 100 protein spots. By<br />
staining with colloidal Coomassie blue about 50 protein<br />
spots were visualized of which 41 were identified<br />
by determining the mass and the partial sequence of<br />
their tryptic peptides following enzymatic digestion.<br />
The identified proteins belonged to several functional<br />
categories, mainly energy metabolism, translation and<br />
heat shock response. In addition, we found lipoproteins<br />
and most of the proteins involved in cytadherence<br />
which were known to be components of the<br />
Triton X-100 insoluble fraction based on evidence<br />
from previous experiments. There were also 11 functionally<br />
unassigned proteins. The quantitatively most<br />
prevalent proteins were the heat shock protein DnaK,<br />
the elongation factor Tu and the subunits α and ß<br />
of the pyruvate dehydrogenase (PdhA, PdhB). To<br />
prove whether a cytoskeleton-like structure exists<br />
in Mycoplasma pneumoniae, further experiments are<br />
required.<br />
One of the promising newer methods to identify in<br />
vivo interacting proteins is the two-hybrid system in<br />
Saccharomyces cerevisiae. We started in cooperation<br />
with M. Kögel (LION Bioscience, Heidelberg) a twohybrid<br />
analysis with the aim of finding which of the<br />
proposed proteins of the cytoskeleton-like structure<br />
interact directly. We expect that this approach will<br />
also reveal new candidates for structural components.<br />
As starting material for our analysis we used the gene<br />
hmw2. Our selection is based on data from various<br />
laboratories indicating that the gene hmw2 codes for<br />
one of the key components in the formation of the<br />
cytoskeleton-like structures. So far, we have identified<br />
several interacting proteins. These results are now<br />
being verified by a second independent method.<br />
77