ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
ZMBH J.Bericht 2000 - Zentrum für Molekulare Biologie der ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
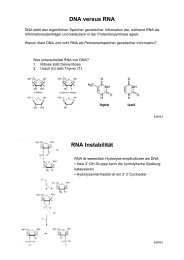
Aß42/Aß40 ratios (Figure 2). A massive effect was<br />
observed for A4CT-I45F (34-fold increase) making<br />
this construct important for the generation of animal<br />
models for Alzheimer‘s disease. Unlike the other mutations,<br />
A4CT-V44F was processed mainly to Aß38, as<br />
determined by mass spectrometry. Our data provide a<br />
detailed model for the active site of γ-secretase (Figure<br />
2). According to this model Aß40 is produced when<br />
γ-secretase interacts with APP by binding to one side<br />
of the alpha-helical transmembrane domain of APP.<br />
Alternatively, Aß42 arises by binding of γ-secretase<br />
to the opposite side (Fig. 2). Mutations in the transmembrane<br />
domain of APP interfere with the interaction<br />
between γ-secretase and APP and, thus, alter the<br />
cleavage specificity of γ-secretase (Fig. 2).<br />
Figure 2: Schematic representation of the amino acid positions<br />
(P) of the ß-secretase product A4CT of APP relative to<br />
the cleavage site of γ-secretase. Top: linear arrangement of the<br />
residues relative to the cleavage site after residue 40 (Aß40)<br />
and residue 42 (Aß42). The scissile peptide bond is constituted<br />
by residues P1 and P1‘. Bottom: helical wheel arrangement<br />
of amino acids 40 to 49 of A4CT with respect to the cleavage<br />
sites after residues 40 and 42. Binding of γ-secretase to the<br />
transmembrane domain giving rise to Aß40 or Aß42 has to<br />
occur at opposite sides of the helix.<br />
28<br />
IV. Regulation of exon 15 splicing of APP<br />
premRNA<br />
C. Bergsdorff, S. Kreger, K. Paliga<br />
Alternative splicing of exon 15 of the amyloid precursor<br />
protein (APP) pre-mRNA generates two APP<br />
isoform groups APP(ex15) (containing exon 15) and<br />
L-APP (without exon 15), which show a cell-specific<br />
distribution in non-neuronal cells and neurons of rat.<br />
Both APP isoforms differ in regard to functional properties<br />
like post-translational modification, APP secretion,<br />
and proteolytic production of Aß peptide from<br />
APP molecules. Since Aß generation is an important<br />
factor in the development of Alzheimer‘s disease, one<br />
could anticipate that these major APP isoforms might<br />
contribute differentially to the mechanisms un<strong>der</strong>lying<br />
neurodegeneration in Alzheimer‘s disease. We<br />
established an APP minigene system in a murine cell<br />
system to identify cis-acting elements controlling exon<br />
15 recognition. A 12.5-kilobase pair genomic fragment<br />
of the murine APP gene contained all cis-regulatory<br />
elements to reproduce the splicing pattern of the<br />
endogenous APP transcripts. By using this approach,<br />
two intronic cis-elements flanking exon 15 were identified<br />
that block the inclusion of exon 15 in APP transcripts<br />
of non-neuronal cells. Point mutation analysis<br />
of these intronic regions indicated that pyrimidinerich<br />
sequences are involved in the splice repressor<br />
function. Finally, grafting experiments demonstrated<br />
that these regulatory regions cell-specifically enhance<br />
the blockage of a chimeric exon in the non-neuronal<br />
splicing system.<br />
V. Physiological function of APP<br />
A. Fossgreen, F. Reinhard, S. Scheurermann, P.<br />
Soba, J. Stumm<br />
In collaboration with B. Brückner and R. Paro, <strong>ZMBH</strong>,<br />
we used Drosophila melanogaster as a model system<br />
to analyze the function of APP by expressing wildtype<br />
and various mutant forms of human APP in fly<br />
tissue culture cells as well as in transgenic fly lines.<br />
After expression of full-length APP forms, secretion<br />
of APP but not of Aß was observed in both systems. By<br />
using SPA4CT, a short APP form in which the signal<br />
peptide was fused directly to the Aß region, transmembrane<br />
domain, and cytoplasmic tail, we observed<br />
Aß release in flies and fly-tissue culture cells. Consequently,<br />
we showed a γ-secretase activity in flies.<br />
Interestingly, transgenic flies expressing full-length<br />
forms of APP have a blistered-wing phenotype. As<br />
the wing is composed of interacting dorsal and ventral<br />
epithelial cell layers, this phenotype suggests that<br />
human APP expression interferes with cell adhesion/<br />
signaling pathways in Drosophila, independently of<br />
Aß generation. Wing morphogenesis in Drosophila is<br />
characterized by the apposition of two epithelial cell<br />
layers, the dorsal and ventral layer and may thus be<br />
used as a model to analyze the role of APP in the interaction<br />
of presynaptic and postsynaptic membranes at<br />
the synapse. We presume that APP negatively interacts<br />
with factors involved in the adhesion of the two wing<br />
epithelia and the two synaptic membranes, respectively.<br />
In Drosophila, some mutations deficient for the expression<br />
of specific integrin subunits develop distinct wing<br />
blisters whereas another one was shown to be involved<br />
in short term memory. This provides an interesting<br />
link between physiological APP functions, integrinmediated<br />
adhesion processes and memory mechanisms.<br />
Since integrins are heterodimeric receptors that<br />
may be ligand-bound to extracellular matrix molecules<br />
and may have a signaling function, integrins are<br />
especially required for the function of the cell-matrixcell<br />
junction, where one surface of a cell such as a<br />
synaptic density adheres to the other. This function<br />
of integrins supports our suggestion of APP’s involvement<br />
in cell adhesion. We envisage that APP might<br />
act as an antagonist for integrins, both in the developing<br />
wing and at plasticity of the synapse. APP could<br />
bind, either directly or via other molecules to the same<br />
extracellular matrix sites as integrins. Indeed, in rat<br />
primary neurons APP and integrins were colocalized<br />
in structures that are reminiscent of synapses. However,<br />
APP might be involved in other pathways resulting<br />
in a deterioration of the adhesion or signaling<br />
pathways supported by the integrins. For example,<br />
expression of a mutant form of G protein involved in<br />
cAMP signaling or the blistered gene encoding a protein<br />
related to human serum response factors result<br />
in Drosophila a comparable wing. Taken together,<br />
Drosophila seems to be a valuable animal-model<br />
system for studying the physiological and pathogenic<br />
functions of human APP since it may be suited for<br />
unraveling APP transmembrane signaling mechanisms<br />
potentially related to Alzheimer‘s disease. By using<br />
this fly model, testing genetic interactions between<br />
APP-expressing lines producing the blistered phenotype<br />
and other mutants engaged in wing morphology<br />
offers the perspective to unravel those physiological<br />
and pathogenic pathways linked to human APP.<br />
VI. Regulation of APP-Aß metabolism by cholesterol<br />
C. Bergmann, T. Hartmann, H. Runz, P. Semenfeld,<br />
I. Tomic<br />
Cholesterol metabolism and Alzheimer‘s disease are<br />
genetically linked. The apoEε4 allele of the apolipoprotein<br />
E gene is associated with higher cholesterol<br />
levels and was shown to increase the risk of develop-<br />
29