linea guida aiom - terapia del dolore in oncologia - Azienda USL di ...
linea guida aiom - terapia del dolore in oncologia - Azienda USL di ...
linea guida aiom - terapia del dolore in oncologia - Azienda USL di ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>in</strong> <strong>terapia</strong> e un controllo <strong>in</strong>sufficiente <strong>del</strong> <strong>dolore</strong>. E’ opportuno che questa procedura sia<br />
effettuata <strong>in</strong> collaborazione con uno specialista <strong>di</strong> cure palliative o <strong>del</strong>la <strong>terapia</strong> <strong>del</strong> <strong>dolore</strong><br />
Raccomandazione Positiva Debole<br />
Bibliografia<br />
1-Quigley C. Opioid switch<strong>in</strong>g to improve pa<strong>in</strong> relief and drug tolerability.Cochrane Database Syst<br />
Rev. 2004;(3):CD004847. Review. PubMed PMID: 15266542.<br />
2-Mercadante S, Ferrera P, Villari P et al. Frequency, <strong>in</strong><strong>di</strong>cations, outcomes and pre<strong>di</strong>ctive factors<br />
of opioid switch<strong>in</strong>g <strong>in</strong> an acute palliative care unit. J Pa<strong>in</strong> Sympt Manage, 2009, 37:632-641.<br />
3-Mercadante S., Bruera E. Opioid switch<strong>in</strong>g: a systematic and critical review. Cancer Treatment<br />
Review, 2006, 32:304-315.<br />
4-Riley J, Ross JR, Rutter D et al. No pa<strong>in</strong> relief from morph<strong>in</strong>e? In<strong>di</strong>vidual variation <strong>in</strong> sensitivity<br />
to morph<strong>in</strong>e and the need to switch to an alternative opioid <strong>in</strong> cancer patients. Supp Care Cancer,<br />
2006, 14:56-64.<br />
5-Muller-Busch HC., L<strong>in</strong>dena G., Tietze K. et al. Opioid switch <strong>in</strong> palliative care, opioid choice by<br />
cl<strong>in</strong>ical need and opioid availability. Eur J Pa<strong>in</strong> 2005; 9:571-579<br />
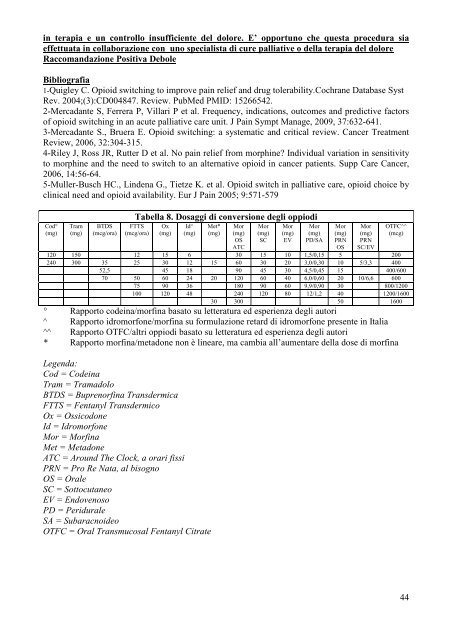
Cod°<br />
(mg)<br />
Tram<br />
(mg)<br />
BTDS<br />
(mcg/ora)<br />
Tabella 8. Dosaggi <strong>di</strong> conversione degli oppio<strong>di</strong><br />
FTTS<br />
(mcg/ora)<br />
Ox<br />
(mg)<br />
Id^<br />
(mg)<br />
Met*<br />
(mg)<br />
Mor<br />
(mg)<br />
OS<br />
ATC<br />
Mor<br />
(mg)<br />
SC<br />
Mor<br />
(mg)<br />
EV<br />
Mor<br />
(mg)<br />
PD/SA<br />
Mor<br />
(mg)<br />
PRN<br />
OS<br />
Mor<br />
(mg)<br />
PRN<br />
SC/EV<br />
OTFC^^<br />
(mcg)<br />
120 150 12 15 6 30 15 10 1,5/0,15 5 200<br />
240 300 35 25 30 12 15 60 30 20 3,0/0,30 10 5/3,3 400<br />
52,5 45 18 90 45 30 4,5/0,45 15 400/600<br />
70 50 60 24 20 120 60 40 6,0/0,60 20 10/6,6 600<br />
75 90 36 180 90 60 9,9/0,90 30 800/1200<br />
100 120 48 240 120 80 12/1,2 40 1200/1600<br />
30 300 50 1600<br />
° Rapporto code<strong>in</strong>a/morf<strong>in</strong>a basato su letteratura ed esperienza degli autori<br />
^ Rapporto idromorfone/morf<strong>in</strong>a su formulazione retard <strong>di</strong> idromorfone presente <strong>in</strong> Italia<br />
^^ Rapporto OTFC/altri oppio<strong>di</strong> basato su letteratura ed esperienza degli autori<br />
* Rapporto morf<strong>in</strong>a/metadone non è <strong>l<strong>in</strong>ea</strong>re, ma cambia all’aumentare <strong>del</strong>la dose <strong>di</strong> morf<strong>in</strong>a<br />
Legenda:<br />
Cod = Code<strong>in</strong>a<br />
Tram = Tramadolo<br />
BTDS = Buprenorf<strong>in</strong>a Transdermica<br />
FTTS = Fentanyl Transdermico<br />
Ox = Ossicodone<br />
Id = Idromorfone<br />
Mor = Morf<strong>in</strong>a<br />
Met = Metadone<br />
ATC = Around The Clock, a orari fissi<br />
PRN = Pro Re Nata, al bisogno<br />
OS = Orale<br />
SC = Sottocutaneo<br />
EV = Endovenoso<br />
PD = Peridurale<br />
SA = Subaracnoideo<br />
OTFC = Oral Transmucosal Fentanyl Citrate<br />
44