Self-Assembly of Synthetic and Biological Polymeric Systems of ...
Self-Assembly of Synthetic and Biological Polymeric Systems of ...
Self-Assembly of Synthetic and Biological Polymeric Systems of ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
One-Dimensional Magnetic Nanowires<br />
(spin–spin) relaxation process <strong>of</strong> the interaction between the<br />
excited nuclei <strong>and</strong> those with lower energy. The efficiency <strong>of</strong><br />
an MRI contrast agent is commonly assessed in terms <strong>of</strong> its<br />
relaxivities (r 1 <strong>and</strong> r 2), which are rates <strong>of</strong> proton relaxation<br />
determined by Equation (1):<br />
1=T i,obs ¼ 1=T i,d þ r i½MŠ ðI ¼ 1, 2Þ ð1Þ<br />
in which 1/T i,obs is the observed solvent relaxation rate in the<br />
presence <strong>of</strong> a contrast agent, 1/T i,d is the relaxation rate <strong>of</strong><br />
the pure diamagnetic solvent, <strong>and</strong> [M] is the concentration<br />
<strong>of</strong> the contrast agent. [33] For a T 2 contrast agent, the higher<br />
the r 2*/r 1 ratio the better the contrast efficacy. The spin–spin<br />
<strong>and</strong> spin–lattice relaxation times weighted spin-echo MRI <strong>of</strong><br />
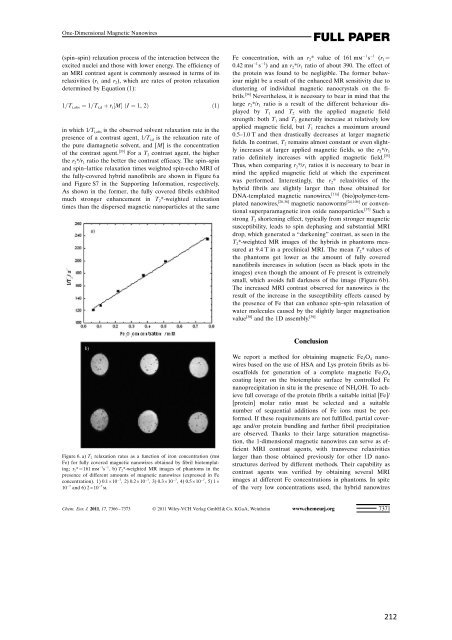
the fully-covered hybrid nan<strong>of</strong>ibrils are shown in Figure 6 a<br />
<strong>and</strong> Figure S7 in the Supporting Information, respectively.<br />
As shown in the former, the fully covered fibrils exhibited<br />
much stronger enhancement in T 2*-weighted relaxation<br />
times than the dispersed magnetic nanoparticles at the same<br />
Figure 6. a) T 2 relaxation rates as a function <strong>of</strong> iron concentration (mm<br />
Fe) for fully covered magnetic nanowires obtained by fibril biotemplating;<br />
r 2*=161 mm 1 s 1 .b)T 2*-weighted MR images <strong>of</strong> phantoms in the<br />
presence <strong>of</strong> different amounts <strong>of</strong> magnetic nanowires (expressed in Fe<br />
concentration). 1) 0.1”10 7 , 2) 0.2”10 7 , 3) 0.3”10 7 , 4) 0.5”10 7 ,5)1”<br />
10 7 <strong>and</strong> 6) 2”10 7 m.<br />
Fe concentration, with an r 2* value <strong>of</strong> 161 mm 1 s 1 (r 1=<br />
0.42 mm 1 s 1 ) <strong>and</strong> an r 2*/r 1 ratio <strong>of</strong> about 390. The effect <strong>of</strong><br />
the protein was found to be negligible. The former behaviour<br />
might be a result <strong>of</strong> the enhanced MR sensitivity due to<br />
clustering <strong>of</strong> individual magnetic nanocrystals on the fibrils.<br />
[34] Nevertheless, it is necessary to bear in mind that the<br />
large r 2*/r 1 ratio is a result <strong>of</strong> the different behaviour displayed<br />
by T 1 <strong>and</strong> T 2 with the applied magnetic field<br />
strength: both T 1 <strong>and</strong> T 2 generally increase at relatively low<br />
applied magnetic field, but T 1 reaches a maximum around<br />
0.5–1.0 T <strong>and</strong> then drastically decreases at larger magnetic<br />
fields. In contrast, T 2 remains almost constant or even slightly<br />
increases at larger applied magnetic fields, so the r 2*/r 1<br />
ratio definitely increases with applied magnetic field. [35]<br />
Thus, when comparing r 2*/r 1 ratios it is necessary to bear in<br />
mind the applied magnetic field at which the experiment<br />
was performed. Interestingly, the r 2* relaxivities <strong>of</strong> the<br />
hybrid fibrils are slightly larger than those obtained for<br />
DNA-templated magnetic nanowires, [11a] (bio)polymer-templated<br />
nanowires, [20,36] magnetic nanoworms [2d, 10b] or conventional<br />
superparamagnetic iron oxide nanoparticles. [37] Such a<br />
strong T 2 shortening effect, typically from stronger magnetic<br />
susceptibility, leads to spin dephasing <strong>and</strong> substantial MRI<br />
drop, which generated a “darkening” contrast, as seen in the<br />
T 2*-weighted MR images <strong>of</strong> the hybrids in phantoms measured<br />
at 9.4 T in a preclinical MRI. The mean T 2* values <strong>of</strong><br />
the phantoms get lower as the amount <strong>of</strong> fully covered<br />
nan<strong>of</strong>ibrils increases in solution (seen as black spots in the<br />
images) even though the amount <strong>of</strong> Fe present is extremely<br />
small, which avoids full darkness <strong>of</strong> the image (Figure 6 b).<br />
The increased MRI contrast observed for nanowires is the<br />
result <strong>of</strong> the increase in the susceptibility effects caused by<br />
the presence <strong>of</strong> Fe that can enhance spin–spin relaxation <strong>of</strong><br />
water molecules caused by the slightly larger magnetisation<br />
value [38] <strong>and</strong> the 1D assembly. [39]<br />
Conclusion<br />
FULL PAPER<br />
We report a method for obtaining magnetic Fe 3O 4 nanowires<br />
based on the use <strong>of</strong> HSA <strong>and</strong> Lys protein fibrils as bioscaffolds<br />
for generation <strong>of</strong> a complete magnetic Fe 3O 4<br />
coating layer on the biotemplate surface by controlled Fe<br />
nanoprecipitation in situ in the presence <strong>of</strong> NH 4OH. To achieve<br />
full coverage <strong>of</strong> the protein fibrils a suitable initial [Fe]/ACHTUNG-<br />
TRENUNG[protein] molar ratio must be selected <strong>and</strong> a suitable<br />
number <strong>of</strong> sequential additions <strong>of</strong> Fe ions must be performed.<br />
If these requirements are not fulfilled, partial coverage<br />
<strong>and</strong>/or protein bundling <strong>and</strong> further fibril precipitation<br />
are observed. Thanks to their large saturation magnetisation,<br />
the 1-dimensional magnetic nanowires can serve as efficient<br />
MRI contrast agents, with transverse relaxivities<br />
larger than those obtained previously for other 1D nanostructures<br />
derived by different methods. Their capability as<br />
contrast agents was verified by obtaining several MRI<br />
images at different Fe concentrations in phantoms. In spite<br />
<strong>of</strong> the very low concentrations used, the hybrid nanowires<br />
Chem. Eur. J. 2011, 17, 7366 – 7373 2011 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim www.chemeurj.org 7371