Pharmaceutical Technology: Controlled Drug Release, Volume 2
Pharmaceutical Technology: Controlled Drug Release, Volume 2
Pharmaceutical Technology: Controlled Drug Release, Volume 2
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
26 MATRIX FORMULATIONS [CH. 2<br />
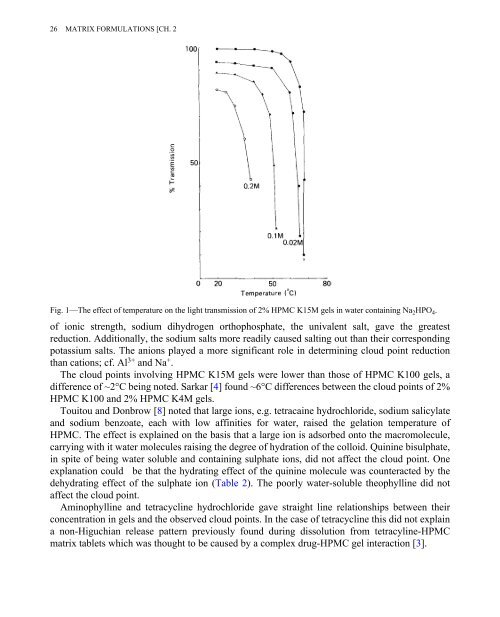
Fig. 1—The effect of temperature on the light transmission of 2% HPMC K15M gels in water containing Na 2 HPO 4 .<br />
of ionic strength, sodium dihydrogen orthophosphate, the univalent salt, gave the greatest<br />
reduction. Additionally, the sodium salts more readily caused salting out than their corresponding<br />
potassium salts. The anions played a more significant role in determining cloud point reduction<br />
than cations; cf. Al 3+ and Na + .<br />
The cloud points involving HPMC K15M gels were lower than those of HPMC K100 gels, a<br />
difference of ~2°C being noted. Sarkar [4] found ~6°C differences between the cloud points of 2%<br />
HPMC K100 and 2% HPMC K4M gels.<br />
Touitou and Donbrow [8] noted that large ions, e.g. tetracaine hydrochloride, sodium salicylate<br />
and sodium benzoate, each with low affinities for water, raised the gelation temperature of<br />
HPMC. The effect is explained on the basis that a large ion is adsorbed onto the macromolecule,<br />
carrying with it water molecules raising the degree of hydration of the colloid. Quinine bisulphate,<br />
in spite of being water soluble and containing sulphate ions, did not affect the cloud point. One<br />
explanation could be that the hydrating effect of the quinine molecule was counteracted by the<br />
dehydrating effect of the sulphate ion (Table 2). The poorly water-soluble theophylline did not<br />
affect the cloud point.<br />
Aminophylline and tetracycline hydrochloride gave straight line relationships between their<br />
concentration in gels and the observed cloud points. In the case of tetracycline this did not explain<br />
a non-Higuchian release pattern previously found during dissolution from tetracyline-HPMC<br />
matrix tablets which was thought to be caused by a complex drug-HPMC gel interaction [3].