Pharmaceutical Technology: Controlled Drug Release, Volume 2
Pharmaceutical Technology: Controlled Drug Release, Volume 2
Pharmaceutical Technology: Controlled Drug Release, Volume 2
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
CH. 5] AN OPTIMIZED PROLONGED RELEASE FORMULATION 61<br />
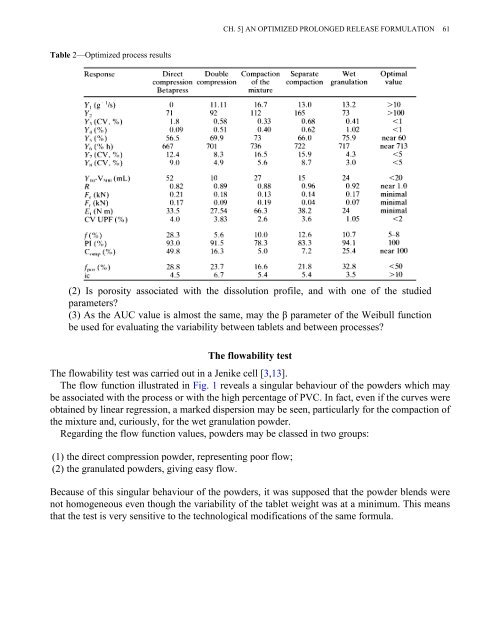
Table 2—Optimized process results<br />
(2) Is porosity associated with the dissolution profile, and with one of the studied<br />
parameters?<br />
(3) As the AUC value is almost the same, may the β parameter of the Weibull function<br />
be used for evaluating the variability between tablets and between processes?<br />
The flowability test<br />
The flowability test was carried out in a Jenike cell [3,13].<br />
The flow function illustrated in Fig. 1 reveals a singular behaviour of the powders which may<br />
be associated with the process or with the high percentage of PVC. In fact, even if the curves were<br />
obtained by linear regression, a marked dispersion may be seen, particularly for the compaction of<br />
the mixture and, curiously, for the wet granulation powder.<br />
Regarding the flow function values, powders may be classed in two groups:<br />
(1) the direct compression powder, representing poor flow;<br />
(2) the granulated powders, giving easy flow.<br />
Because of this singular behaviour of the powders, it was supposed that the powder blends were<br />
not homogeneous even though the variability of the tablet weight was at a minimum. This means<br />
that the test is very sensitive to the technological modifications of the same formula.