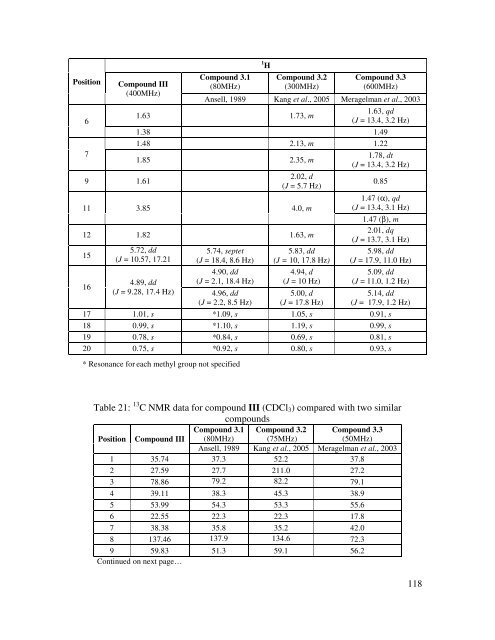
Position Moiety Compound III (400MHz) 11 CH-OH 3.85, m 1 H Ansell, 1989 (80MHz) 3.86, ddd (J = 4.7, 7.1, 11.7 Hz) Compound III (400MHz) 13 C # Ansell, 1989 (20MHz) 66.19 69.3 12 CH2 1.82 (α) 1.35 (β) 45.85 40.7 *13 C 39.57 38.7 14 CH= 5.20, s 5.21, d (J = 1.5 Hz) 127.64 128.4 15 CH= 5.72, dd (J = 10.57, 17.21 Hz) 5.74, sx (J = 9.5, 18.1 Hz) 4.87, dd 146.18 145.4 16 CH2 4.89, dd (J = 9.28, 17.4 Hz) (J = 1.9, 18.1 Hz) 4.91, dd (J = 1.9, 9.5 Hz) 112.69 112.5 17 CH3 1.01, s 1.02, s 29.51 29.5 18 CH3 0.99, s 1.00, s 28.43 28.3 19 CH3 0.78, s 0.79, s 15.59 15.7 20 CH3 0.75, s 0.76, s 15.82 16.6 3 OCOCH3 --- --- --- 170.2 3 OCOCH3 --- --- --- 21.1 11 OCOCH3 --- --- --- 170.6 11 OCOCH3 --- --- --- 21.5 * these resonances can be used interchangeably # The 13 C NMR data corresponds to the acetylated product <strong>of</strong> compound III Position 1 2 3 Table 20: 1 H NMR data for compound III (CDCl3) compared with two similar compounds Compound III (400MHz) Compound 3.1 (80MHz) 1 H Compound 3.2 (300MHz) Compound 3.3 (600MHz) Ansell, 1989 Kang et al., 2005 Meragelman et al., 2003 2.35 2.68, s 1.71, dt (J = 13.1, 3.5 Hz) 2.02 0.98 1.69 1.61, m 1.57 1.61, m 3.26, dd (J = 3.84, 10.84 Hz) 3.31, dd (J = 4.5, 9.6 Hz) 4.0, s 3.21, dd (J = 11.1, 5,2 Hz) 5 1.08 1.68, m 0.82 Continued on next page…. 117
Position 6 7 Compound III (400MHz) 9 1.61 Compound 3.1 (80MHz) 1 H Compound 3.2 (300MHz) Compound 3.3 (600MHz) Ansell, 1989 Kang et al., 2005 Meragelman et al., 2003 1.63 1.73, m 1.63, qd (J = 13.4, 3.2 Hz) 1.38 1.49 1.48 2.13, m 1.22 1.85 2.35, m 2.02, d (J = 5.7 Hz) 11 3.85 4.0, m 12 1.82 1.63, m 15 16 5.72, dd (J = 10.57, 17.21 Hz) 4.89, dd (J = 9.28, 17.4 Hz) 5.74, septet (J = 18.4, 8.6 Hz) 4.90, dd (J = 2.1, 18.4 Hz) 4.96, dd (J = 2.2, 8.5 Hz) 5.83, dd (J = 10, 17.8 Hz) 4.94, d (J = 10 Hz) 5.00, d (J = 17.8 Hz) 1.78, dt (J = 13.4, 3.2 Hz) 0.85 1.47 (α), qd (J = 13.4, 3.1 Hz) 1.47 (β), m 2.01, dq (J = 13.7, 3.1 Hz) 5.98, dd (J = 17.9, 11.0 Hz) 5.09, dd (J = 11.0, 1.2 Hz) 5.14, dd (J = 17.9, 1.2 Hz) 17 1.01, s *1.09, s 1.05, s 0.91, s 18 0.99, s *1.10, s 1.19, s 0.99, s 19 0.78, s *0.84, s 0.69, s 0.81, s 20 0.75, s *0.92, s 0.80, s 0.93, s * Resonance for each methyl group not specified Table 21: 13 C NMR data for compound III (CDCl3) compared with two similar compounds Compound 3.1 Compound 3.2 Compound 3.3 Position Compound III (80MHz) (75MHz) (50MHz) Ansell, 1989 Kang et al., 2005 Meragelman et al., 2003 1 35.74 37.3 52.2 37.8 2 27.59 27.7 211.0 27.2 3 78.86 79.2 82.2 79.1 4 39.11 38.3 45.3 38.9 5 53.99 54.3 53.3 55.6 6 22.55 22.3 22.3 17.8 7 38.38 35.8 35.2 42.0 8 137.46 137.9 134.6 72.3 9 59.83 51.3 59.1 56.2 Continued on next page… 118
- Page 1 and 2:
UNIVERSITY OF KWAZULU-NATAL FACULTY
- Page 3 and 4:
Preface The experimental work descr
- Page 5 and 6:
Acknowledgements First and foremost
- Page 7 and 8:
GC-MS - gas chromatography-mass spe
- Page 9 and 10:
Table 19: NMR data of compound III
- Page 11 and 12:
Spectrum 1d: Expanded DEPT spectrum
- Page 13 and 14:
Compound V, stigmasterol 217 Spectr
- Page 15 and 16:
2 19 1 4 18 O 20 5 Structures of co
- Page 17 and 18:
3.4 Results and Discussion of the c
- Page 19 and 20:
compounds are also classified as tr
- Page 21 and 22:
1 H CO2H 19 20 CHO 18 10 5 H 12 14
- Page 23 and 24:
may be referred to as a norabietane
- Page 25 and 26:
In the mevalonic acid pathway, the
- Page 27 and 28:
N thiamine diphosphate (TPP) O OH N
- Page 29 and 30:
in the kauranes. From the kauranes,
- Page 31 and 32:
References Abad, A., Agullo, C., Cu
- Page 33 and 34:
The flower colour varies among the
- Page 35 and 36:
Clade 1 unplaced groups Group A: Pl
- Page 37 and 38:
different medical conditions and in
- Page 39 and 40:
Plectranthus species Skin condition
- Page 41 and 42:
Plectranthus species Heart, circula
- Page 43 and 44:
Use of the minimum inhibitory conce
- Page 45 and 46:
Plectranthus species Coleus kiliman
- Page 47 and 48:
Plectranthus species Extraction med
- Page 49 and 50:
Plectranthus species Coleus kiliman
- Page 51 and 52:
Methanol, ethanol and water seem to
- Page 53 and 54:
Plectranthus species P. amboinicus
- Page 55 and 56:
Plectranthus species P. amboinicus
- Page 57 and 58:
variation is observed within ring B
- Page 59 and 60:
Compound Name Synonym 39 40 41 42 4
- Page 61 and 62:
compounds (Table 6b). Depending on
- Page 63 and 64:
Table 6c: Royleanone-type abietanes
- Page 65 and 66:
R 1 R2CH2 19 O H 3CCO 19 O H 6 12 O
- Page 67 and 68:
Compound Name Synonym 81 82 83 84 8
- Page 69 and 70:
Compound Name Synonym 104 105 106 1
- Page 71 and 72:
R 1 19 3 3 1 18 1 18 20 20 O H O H
- Page 73 and 74:
educed furan ring. The linear side
- Page 75 and 76:
Compound Name Synonym HCO O 128 129
- Page 77 and 78:
Thirty-six compounds (131-167) havi
- Page 79 and 80:
Compound Name Synonym 147 148 HO HO
- Page 81 and 82:
In compounds 149-165 (Table 9b), an
- Page 83 and 84: Compound Name Synonym 163 164 165 R
- Page 85 and 86: Table 9c: Coleon-type abietanes wit
- Page 87 and 88: 1 18 3 20 4 O 5 19 R 1 R2 11 9 10 A
- Page 89 and 90: The majority of the abietanes isola
- Page 91 and 92: Plectranthus species Plant part Com
- Page 93 and 94: The leaves seem to be the most freq
- Page 95 and 96: Table 14 contains six abietanes iso
- Page 97 and 98: References Abdel-Mogib, M., Albar,
- Page 99 and 100: Cos, P., Hermans, N., de Bryne, T.,
- Page 101 and 102: Hulme, M. (1954) Wild flowers of Na
- Page 103 and 104: Lingling, X., Jie, L., Weijia, L. a
- Page 105 and 106: Oliveira, P.M., Alves, R.B., Ferrei
- Page 107 and 108: Ruedi, P. and Eugster, C. H. (1977)
- Page 109 and 110: Ulubelen, A., Birman, H., Oksuz, S.
- Page 111 and 112: Chapter 3 Extractives from Plectran
- Page 113 and 114: 4 20 10 O H 19 18 6 7 OH OH C 13 R
- Page 115 and 116: a few cases (specifically stated in
- Page 117 and 118: The leaves (184.63g) were air-dried
- Page 119 and 120: The ethyl acetate extract (0.98g) w
- Page 121 and 122: 3.4.1 Structural elucidation of Com
- Page 123 and 124: esonance showed HMBC correlations t
- Page 125 and 126: esonance at δC 185.76 of C-14 is a
- Page 127 and 128: Position Moiety Compound I 3 CH2 5
- Page 129 and 130: Table 18: 13 C NMR data for compoun
- Page 131 and 132: The equatorial methyl group at C-4,
- Page 133: The proton NMR data for compound II
- Page 137 and 138: 3.4.3 Structure elucidation of Comp
- Page 139 and 140: showed HMBC correlations to another
- Page 141 and 142: H HO 3 OH H 2 4 24 eq 1 23 5 ax Hax
- Page 143 and 144: HO HO Position 2 23 4 10 24 25 26 9
- Page 145 and 146: Position Compound IV 4.1 4.2 4.3 b
- Page 147 and 148: multiplet at δH 1.91 (H-21) and a
- Page 149 and 150: 1 H Position Compound VI Burns et a
- Page 151 and 152: Ultraviolet spectrum (λ max): 192n
- Page 153 and 154: References Ansell, S.M. (1989) The
- Page 155 and 156: Laing, M.D., Drewes, S.E. and Gurla
- Page 157 and 158: van Vuuren, S. (2007) The antimicro
- Page 159 and 160: Since the 7α-isomers of compounds
- Page 161 and 162: eing incubated for 24 hours at 37°
- Page 163 and 164: This indicated that the dihydroxy c
- Page 165 and 166: acetic acid and air drying the plat
- Page 167 and 168: whereas GI50 values for the renal a
- Page 169 and 170: Table 28: Growth inhibition values
- Page 171 and 172: Gaspar-Marques, C., Duarte, M.A., S
- Page 173 and 174: Chapter 5 Conclusion The aim of thi
- Page 175 and 176: References Abdel-Mogib, M., Albar,
- Page 177 and 178: Table of Contents Page Compound I,
- Page 179 and 180: Compound II, 6β,7β-dihydroxyroyle
- Page 181 and 182: Compound IV, euscaphic acid HO HO 2
- Page 183: HO Compound VI, lupeol 3 24 25 26 2
- Page 236 and 237:
HO 218
- Page 238 and 239:
HO 220
- Page 240 and 241:
222
- Page 242 and 243:
225
- Page 244 and 245:
227