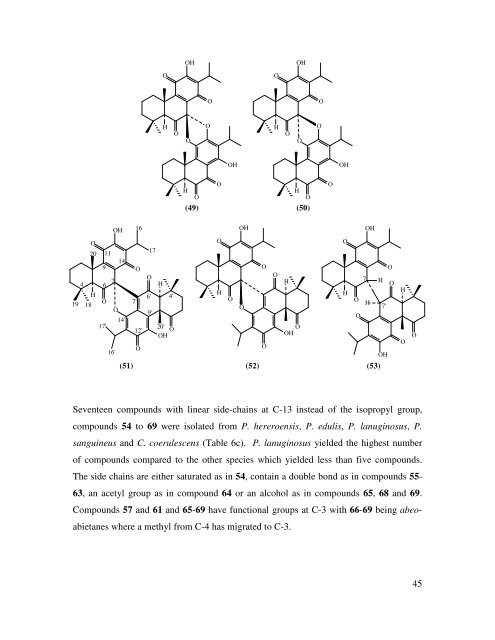
19 O 20 9 11 4 6 7 OH O O H H 18 O O 7' 6' 9' 4' 14' 17' 12' 20' O OH 16' 14 16 O 17 O H O OH O O O OH OH O H H O O (49) (50) O H O OH O O O H O O O OH (51) (52) (53) Seventeen compounds with linear side-chains at C-13 instead <strong>of</strong> the isopropyl group, compounds 54 to 69 were isolated from P. hereroensis, P. edulis, P. lanuginosus, P. sanguineus <strong>and</strong> C. coerulescens (Table 6c). P. lanuginosus yielded the highest number <strong>of</strong> compounds compared to the other species which yielded less than five compounds. The side chains are either saturated as in 54, contain a double bond as in compounds 55- 63, an acetyl group as in compound 64 or an alcohol as in compounds 65, 68 <strong>and</strong> 69. Compounds 57 <strong>and</strong> 61 <strong>and</strong> 65-69 have functional groups at C-3 with 66-69 being abeo- abietanes where a methyl from C-4 has migrated to C-3. O O H O O O OH O H O O 7 OH H H 7' OH O O H O O 45
Table 6c: Royleanone-type abietanes with a linear side chain at C-13 isolated from Plectranthus <strong>and</strong> Coleus species Compound Name Synonym 54 55 56 57 58 59 60 61 62 63 64 Continued on next page…. 16-acetoxy-7α,12-dihydroxy- 8,12-abietadiene-11,14-dione 7α,12-dihydroxy- 17(15→16)-abeoabieta- 8,12,16-triene-11,14-dione 6β,7α,12-trihydroxy- 17(15→16)-abeoabieta- 8,12,16-triene-11,14-dione 3α-formyloxy-6β,7α,12trihydroxy-17(15→16)abeoabieta-8,12,16-triene- 11,14-dione 19-formyloxy-6β,7α,12trihydroxy-17(15→16)abeoabieta-8,12,16-triene- 11,14-dione 7α-ethoxy-6β,12,19trihydroxy-17(15→16)abeoabieta-8,12,16-triene- 11,14-dione 12-hydroxy-17(15→16)abeoabieta-6,8,12,16- tetraene-11,14-dione 3α-formyloxy-12-hydroxy- 17(15→16)-abeoabieta- 6,8,12,16-tetraene-11,14- dione 12,19-dihydroxy- 17(15→16)-abeoabieta- 6,8,12,16-tetraene-11,14- dione 12-hydroxy-19-formyloxy- 17(15→16)-abeoabieta- 6,8,12,16-tetraene-11,14dione 16-acetoxy-7α,12-dihydroxy- 8,12-abietadiene-11,14-dione 6β,7α-dihydroxy(allyl) royleanone Lanugone D Lanugone E Lanugone A Lanugone B Lanugone C Isolated from Plectranthus (P) or Coleus (C) species P. hereroensis P. hereroensis P. sanguineus Reference Gaspar-Marques et al., 2006; Batista et al., 1995 Gaspar-Marques et al., 2006; Batista et al., 1994 Matloubi- Moghadam et al., 1987 P. edulis Kunzle et al., 1987 P. lanuginosus Schmid et al., 1982 P. edulis; P. lanuginosus Kunzle et al., 1987; Schmid et al., 1982 P. edulis Kunzle et al., 1987 P. lanuginosus Schmid et al., 1982 P. hereroensis Batista et al., 1995 46
- Page 1 and 2:
UNIVERSITY OF KWAZULU-NATAL FACULTY
- Page 3 and 4:
Preface The experimental work descr
- Page 5 and 6:
Acknowledgements First and foremost
- Page 7 and 8:
GC-MS - gas chromatography-mass spe
- Page 9 and 10:
Table 19: NMR data of compound III
- Page 11 and 12: Spectrum 1d: Expanded DEPT spectrum
- Page 13 and 14: Compound V, stigmasterol 217 Spectr
- Page 15 and 16: 2 19 1 4 18 O 20 5 Structures of co
- Page 17 and 18: 3.4 Results and Discussion of the c
- Page 19 and 20: compounds are also classified as tr
- Page 21 and 22: 1 H CO2H 19 20 CHO 18 10 5 H 12 14
- Page 23 and 24: may be referred to as a norabietane
- Page 25 and 26: In the mevalonic acid pathway, the
- Page 27 and 28: N thiamine diphosphate (TPP) O OH N
- Page 29 and 30: in the kauranes. From the kauranes,
- Page 31 and 32: References Abad, A., Agullo, C., Cu
- Page 33 and 34: The flower colour varies among the
- Page 35 and 36: Clade 1 unplaced groups Group A: Pl
- Page 37 and 38: different medical conditions and in
- Page 39 and 40: Plectranthus species Skin condition
- Page 41 and 42: Plectranthus species Heart, circula
- Page 43 and 44: Use of the minimum inhibitory conce
- Page 45 and 46: Plectranthus species Coleus kiliman
- Page 47 and 48: Plectranthus species Extraction med
- Page 49 and 50: Plectranthus species Coleus kiliman
- Page 51 and 52: Methanol, ethanol and water seem to
- Page 53 and 54: Plectranthus species P. amboinicus
- Page 55 and 56: Plectranthus species P. amboinicus
- Page 57 and 58: variation is observed within ring B
- Page 59 and 60: Compound Name Synonym 39 40 41 42 4
- Page 61: compounds (Table 6b). Depending on
- Page 65 and 66: R 1 R2CH2 19 O H 3CCO 19 O H 6 12 O
- Page 67 and 68: Compound Name Synonym 81 82 83 84 8
- Page 69 and 70: Compound Name Synonym 104 105 106 1
- Page 71 and 72: R 1 19 3 3 1 18 1 18 20 20 O H O H
- Page 73 and 74: educed furan ring. The linear side
- Page 75 and 76: Compound Name Synonym HCO O 128 129
- Page 77 and 78: Thirty-six compounds (131-167) havi
- Page 79 and 80: Compound Name Synonym 147 148 HO HO
- Page 81 and 82: In compounds 149-165 (Table 9b), an
- Page 83 and 84: Compound Name Synonym 163 164 165 R
- Page 85 and 86: Table 9c: Coleon-type abietanes wit
- Page 87 and 88: 1 18 3 20 4 O 5 19 R 1 R2 11 9 10 A
- Page 89 and 90: The majority of the abietanes isola
- Page 91 and 92: Plectranthus species Plant part Com
- Page 93 and 94: The leaves seem to be the most freq
- Page 95 and 96: Table 14 contains six abietanes iso
- Page 97 and 98: References Abdel-Mogib, M., Albar,
- Page 99 and 100: Cos, P., Hermans, N., de Bryne, T.,
- Page 101 and 102: Hulme, M. (1954) Wild flowers of Na
- Page 103 and 104: Lingling, X., Jie, L., Weijia, L. a
- Page 105 and 106: Oliveira, P.M., Alves, R.B., Ferrei
- Page 107 and 108: Ruedi, P. and Eugster, C. H. (1977)
- Page 109 and 110: Ulubelen, A., Birman, H., Oksuz, S.
- Page 111 and 112: Chapter 3 Extractives from Plectran
- Page 113 and 114:
4 20 10 O H 19 18 6 7 OH OH C 13 R
- Page 115 and 116:
a few cases (specifically stated in
- Page 117 and 118:
The leaves (184.63g) were air-dried
- Page 119 and 120:
The ethyl acetate extract (0.98g) w
- Page 121 and 122:
3.4.1 Structural elucidation of Com
- Page 123 and 124:
esonance showed HMBC correlations t
- Page 125 and 126:
esonance at δC 185.76 of C-14 is a
- Page 127 and 128:
Position Moiety Compound I 3 CH2 5
- Page 129 and 130:
Table 18: 13 C NMR data for compoun
- Page 131 and 132:
The equatorial methyl group at C-4,
- Page 133 and 134:
The proton NMR data for compound II
- Page 135 and 136:
Position 6 7 Compound III (400MHz)
- Page 137 and 138:
3.4.3 Structure elucidation of Comp
- Page 139 and 140:
showed HMBC correlations to another
- Page 141 and 142:
H HO 3 OH H 2 4 24 eq 1 23 5 ax Hax
- Page 143 and 144:
HO HO Position 2 23 4 10 24 25 26 9
- Page 145 and 146:
Position Compound IV 4.1 4.2 4.3 b
- Page 147 and 148:
multiplet at δH 1.91 (H-21) and a
- Page 149 and 150:
1 H Position Compound VI Burns et a
- Page 151 and 152:
Ultraviolet spectrum (λ max): 192n
- Page 153 and 154:
References Ansell, S.M. (1989) The
- Page 155 and 156:
Laing, M.D., Drewes, S.E. and Gurla
- Page 157 and 158:
van Vuuren, S. (2007) The antimicro
- Page 159 and 160:
Since the 7α-isomers of compounds
- Page 161 and 162:
eing incubated for 24 hours at 37°
- Page 163 and 164:
This indicated that the dihydroxy c
- Page 165 and 166:
acetic acid and air drying the plat
- Page 167 and 168:
whereas GI50 values for the renal a
- Page 169 and 170:
Table 28: Growth inhibition values
- Page 171 and 172:
Gaspar-Marques, C., Duarte, M.A., S
- Page 173 and 174:
Chapter 5 Conclusion The aim of thi
- Page 175 and 176:
References Abdel-Mogib, M., Albar,
- Page 177 and 178:
Table of Contents Page Compound I,
- Page 179 and 180:
Compound II, 6β,7β-dihydroxyroyle
- Page 181 and 182:
Compound IV, euscaphic acid HO HO 2
- Page 183:
HO Compound VI, lupeol 3 24 25 26 2
- Page 236 and 237:
HO 218
- Page 238 and 239:
HO 220
- Page 240 and 241:
222
- Page 242 and 243:
225
- Page 244 and 245:
227