A systematic review and economic model of the effectiveness and ...
A systematic review and economic model of the effectiveness and ...
A systematic review and economic model of the effectiveness and ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Items (thous<strong>and</strong>s)<br />
14<br />
12<br />
10<br />
8<br />
6<br />
4<br />
2<br />
0<br />
Mar-99<br />
Sep-99<br />
Items NIC (£)<br />
Mar-00<br />
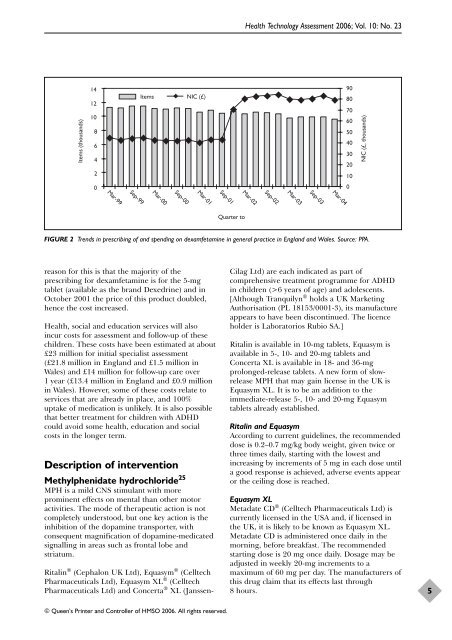
reason for this is that <strong>the</strong> majority <strong>of</strong> <strong>the</strong><br />
prescribing for dexamfetamine is for <strong>the</strong> 5-mg<br />
tablet (available as <strong>the</strong> br<strong>and</strong> Dexedrine) <strong>and</strong> in<br />
October 2001 <strong>the</strong> price <strong>of</strong> this product doubled,<br />
hence <strong>the</strong> cost increased.<br />
Health, social <strong>and</strong> education services will also<br />
incur costs for assessment <strong>and</strong> follow-up <strong>of</strong> <strong>the</strong>se<br />
children. These costs have been estimated at about<br />
£23 million for initial specialist assessment<br />
(£21.8 million in Engl<strong>and</strong> <strong>and</strong> £1.5 million in<br />
Wales) <strong>and</strong> £14 million for follow-up care over<br />
1 year (£13.4 million in Engl<strong>and</strong> <strong>and</strong> £0.9 million<br />
in Wales). However, some <strong>of</strong> <strong>the</strong>se costs relate to<br />
services that are already in place, <strong>and</strong> 100%<br />
uptake <strong>of</strong> medication is unlikely. It is also possible<br />
that better treatment for children with ADHD<br />
could avoid some health, education <strong>and</strong> social<br />
costs in <strong>the</strong> longer term.<br />
Description <strong>of</strong> intervention<br />
Sep-00<br />
Mar-01<br />
Methylphenidate hydrochloride 25<br />
MPH is a mild CNS stimulant with more<br />
prominent effects on mental than o<strong>the</strong>r motor<br />
activities. The mode <strong>of</strong> <strong>the</strong>rapeutic action is not<br />
completely understood, but one key action is <strong>the</strong><br />
inhibition <strong>of</strong> <strong>the</strong> dopamine transporter, with<br />
consequent magnification <strong>of</strong> dopamine-medicated<br />
signalling in areas such as frontal lobe <strong>and</strong><br />
striatum.<br />
Ritalin ® (Cephalon UK Ltd), Equasym ® (Celltech<br />
Pharmaceuticals Ltd), Equasym XL ® (Celltech<br />
Pharmaceuticals Ltd) <strong>and</strong> Concerta ® XL (Janssen-<br />
© Queen’s Printer <strong>and</strong> Controller <strong>of</strong> HMSO 2006. All rights reserved.<br />
Sep-01<br />
Quarter to<br />
Health Technology Assessment 2006; Vol. 10: No. 23<br />
FIGURE 2 Trends in prescribing <strong>of</strong> <strong>and</strong> spending on dexamfetamine in general practice in Engl<strong>and</strong> <strong>and</strong> Wales. Source: PPA.<br />
Mar-02<br />
Sep-02<br />
Mar-03<br />
Sep-03<br />
Mar-04<br />
Cilag Ltd) are each indicated as part <strong>of</strong><br />
comprehensive treatment programme for ADHD<br />
in children (>6 years <strong>of</strong> age) <strong>and</strong> adolescents.<br />
[Although Tranquilyn ® holds a UK Marketing<br />
Authorisation (PL 18153/0001-3), its manufacture<br />
appears to have been discontinued. The licence<br />
holder is Laboratorios Rubio SA.]<br />
Ritalin is available in 10-mg tablets, Equasym is<br />
available in 5-, 10- <strong>and</strong> 20-mg tablets <strong>and</strong><br />
Concerta XL is available in 18- <strong>and</strong> 36-mg<br />
prolonged-release tablets. A new form <strong>of</strong> slowrelease<br />
MPH that may gain license in <strong>the</strong> UK is<br />
Equasym XL. It is to be an addition to <strong>the</strong><br />
immediate-release 5-, 10- <strong>and</strong> 20-mg Equasym<br />
tablets already established.<br />
Ritalin <strong>and</strong> Equasym<br />
According to current guidelines, <strong>the</strong> recommended<br />
dose is 0.2–0.7 mg/kg body weight, given twice or<br />
three times daily, starting with <strong>the</strong> lowest <strong>and</strong><br />
increasing by increments <strong>of</strong> 5 mg in each dose until<br />
a good response is achieved, adverse events appear<br />
or <strong>the</strong> ceiling dose is reached.<br />
Equasym XL<br />
Metadate CD ® (Celltech Pharmaceuticals Ltd) is<br />
currently licensed in <strong>the</strong> USA <strong>and</strong>, if licensed in<br />
<strong>the</strong> UK, it is likely to be known as Equasym XL.<br />
Metadate CD is administered once daily in <strong>the</strong><br />
morning, before breakfast. The recommended<br />
starting dose is 20 mg once daily. Dosage may be<br />
adjusted in weekly 20-mg increments to a<br />
maximum <strong>of</strong> 60 mg per day. The manufacturers <strong>of</strong><br />
this drug claim that its effects last through<br />
8 hours.<br />
90<br />
80<br />
70<br />
60<br />
50<br />
40<br />
30<br />
20<br />
10<br />
0<br />
NIC (£, thous<strong>and</strong>s)<br />
5