Principles of cell signaling - UT Southwestern
Principles of cell signaling - UT Southwestern
Principles of cell signaling - UT Southwestern
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
39057_ch14_<strong>cell</strong>bio.qxd 8/28/06 5:11 PM Page 608<br />
(C)<br />
Catalytic<br />
subunits<br />
PKA<br />
C<br />
R<br />
R<br />
C<br />
Activation <strong>of</strong> PKA by cAMP<br />
(R)<br />
R egulatory<br />
subunits<br />
4 cAMP<br />
R<br />
R<br />
- cAMP<br />
- cAMP<br />
- cAMP<br />
- cAMP<br />
Activated<br />
PKA<br />
C<br />
C<br />
teins throughout the <strong>cell</strong> ranging from ion channels<br />
to transcription factors, and its conserved<br />
substrate preference frequently permits prediction<br />
<strong>of</strong> substrates by sequence analysis. The<br />
cAMP response element binding protein CREB<br />
is phosphorylated by PKA on Ser 133 and is<br />
largely responsible for the impact <strong>of</strong> cAMP on<br />
transcription <strong>of</strong> numerous genes.<br />
R 2 C 2 + 4 cAMP<br />
Kinase activity as a<br />
function <strong>of</strong> [cAMP] (%)<br />
100<br />
80<br />
60<br />
40<br />
20<br />
10%<br />
R 2<br />
. cAMP 4 + 2C<br />
90%<br />
14.15<br />
Ca2+ <strong>signaling</strong> serves<br />
diverse purposes in all<br />
eukaryotic <strong>cell</strong>s<br />
Key concepts<br />
• Ca2+ serves as a second messenger and regulatory<br />
molecule in essentially all <strong>cell</strong>s.<br />
• Ca2+ acts directly on many target proteins and also<br />
regulates the activity <strong>of</strong> a regulatory protein<br />
calmodulin.<br />
• The cytosolic concentration <strong>of</strong> Ca2+ is controlled by<br />
organellar sequestration and release.<br />
2 x 10 -9 2 x 10 -8 2 x 10 -7<br />
[cAMP]<br />
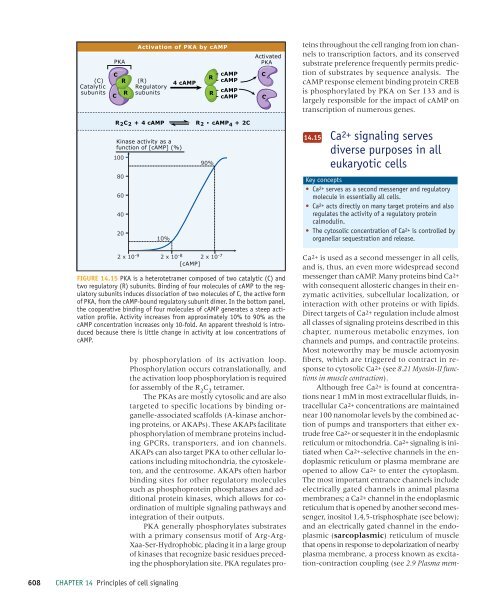
FIGURE 14.15 PKA is a heterotetramer composed <strong>of</strong> two catalytic (C) and<br />
two regulatory (R) subunits. Binding <strong>of</strong> four molecules <strong>of</strong> cAMP to the regulatory<br />
subunits induces dissociation <strong>of</strong> two molecules <strong>of</strong> C, the active form<br />
<strong>of</strong> PKA, from the cAMP-bound regulatory subunit dimer. In the bottom panel,<br />
the cooperative binding <strong>of</strong> four molecules <strong>of</strong> cAMP generates a steep activation<br />
pr<strong>of</strong>ile. Activity increases from approximately 10% to 90% as the<br />
cAMP concentration increases only 10-fold. An apparent threshold is introduced<br />
because there is little change in activity at low concentrations <strong>of</strong><br />
cAMP.<br />
by phosphorylation <strong>of</strong> its activation loop.<br />
Phosphorylation occurs cotranslationally, and<br />
the activation loop phosphorylation is required<br />
for assembly <strong>of</strong> the R 2<br />
C 2<br />
tetramer.<br />
The PKAs are mostly cytosolic and are also<br />
targeted to specific locations by binding organelle-associated<br />
scaffolds (A-kinase anchoring<br />
proteins, or AKAPs). These AKAPs facilitate<br />
phosphorylation <strong>of</strong> membrane proteins including<br />
GPCRs, transporters, and ion channels.<br />
AKAPs can also target PKA to other <strong>cell</strong>ular locations<br />
including mitochondria, the cytoskeleton,<br />
and the centrosome. AKAPs <strong>of</strong>ten harbor<br />
binding sites for other regulatory molecules<br />
such as phosphoprotein phosphatases and additional<br />
protein kinases, which allows for coordination<br />
<strong>of</strong> multiple <strong>signaling</strong> pathways and<br />
integration <strong>of</strong> their outputs.<br />
PKA generally phosphorylates substrates<br />
with a primary consensus motif <strong>of</strong> Arg-Arg-<br />
Xaa-Ser-Hydrophobic, placing it in a large group<br />
<strong>of</strong> kinases that recognize basic residues preceding<br />
the phosphorylation site. PKA regulates pro-<br />
Ca2+ is used as a second messenger in all <strong>cell</strong>s,<br />
and is, thus, an even more widespread second<br />
messenger than cAMP. Many proteins bind Ca2+<br />
with consequent allosteric changes in their enzymatic<br />
activities, sub<strong>cell</strong>ular localization, or<br />
interaction with other proteins or with lipids.<br />
Direct targets <strong>of</strong> Ca2+ regulation include almost<br />
all classes <strong>of</strong> <strong>signaling</strong> proteins described in this<br />
chapter, numerous metabolic enzymes, ion<br />
channels and pumps, and contractile proteins.<br />
Most noteworthy may be muscle actomyosin<br />
fibers, which are triggered to contract in response<br />
to cytosolic Ca2+ (see 8.21 Myosin-II functions<br />
in muscle contraction).<br />
Although free Ca2+ is found at concentrations<br />
near 1 mM in most extra<strong>cell</strong>ular fluids, intra<strong>cell</strong>ular<br />
Ca2+ concentrations are maintained<br />
near 100 nanomolar levels by the combined action<br />
<strong>of</strong> pumps and transporters that either extrude<br />
free Ca2+ or sequester it in the endoplasmic<br />
reticulum or mitochondria. Ca2+ <strong>signaling</strong> is initiated<br />
when Ca2+-selective channels in the endoplasmic<br />
reticulum or plasma membrane are<br />
opened to allow Ca2+ to enter the cytoplasm.<br />
The most important entrance channels include<br />
electrically gated channels in animal plasma<br />
membranes; a Ca2+ channel in the endoplasmic<br />
reticulum that is opened by another second messenger,<br />
inositol 1,4,5-trisphosphate (see below);<br />
and an electrically gated channel in the endoplasmic<br />
(sarcoplasmic) reticulum <strong>of</strong> muscle<br />
that opens in response to depolarization <strong>of</strong> nearby<br />
plasma membrane, a process known as excitation-contraction<br />
coupling (see 2.9 Plasma mem-<br />
608 CHAPTER 14 <strong>Principles</strong> <strong>of</strong> <strong>cell</strong> <strong>signaling</strong>