View - DSpace UniPR
View - DSpace UniPR
View - DSpace UniPR
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Arginine-PNAs<br />
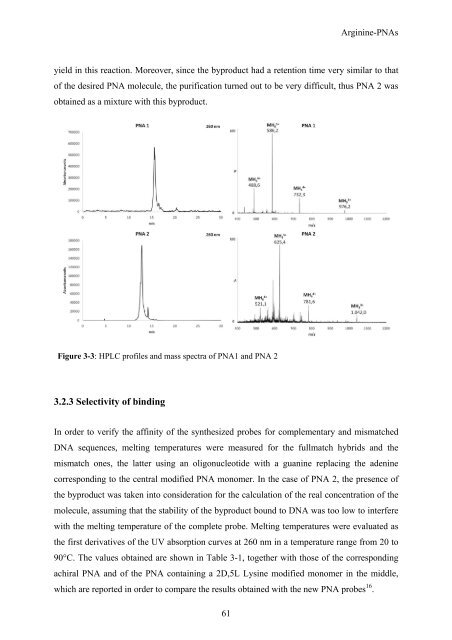
yield in this reaction. Moreover, since the byproduct had a retention time very similar to that<br />
of the desired PNA molecule, the purification turned out to be very difficult, thus PNA 2 was<br />
obtained as a mixture with this byproduct.<br />
Figure 3-3: HPLC profiles and mass spectra of PNA1 and PNA 2<br />
3.2.3 Selectivity of binding<br />
In order to verify the affinity of the synthesized probes for complementary and mismatched<br />
DNA sequences, melting temperatures were measured for the fullmatch hybrids and the<br />
mismatch ones, the latter using an oligonucleotide with a guanine replacing the adenine<br />
corresponding to the central modified PNA monomer. In the case of PNA 2, the presence of<br />
the byproduct was taken into consideration for the calculation of the real concentration of the<br />
molecule, assuming that the stability of the byproduct bound to DNA was too low to interfere<br />
with the melting temperature of the complete probe. Melting temperatures were evaluated as<br />
the first derivatives of the UV absorption curves at 260 nm in a temperature range from 20 to<br />
90°C. The values obtained are shown in Table 3-1, together with those of the corresponding<br />
achiral PNA and of the PNA containing a 2D,5L Lysine modified monomer in the middle,<br />
which are reported in order to compare the results obtained with the new PNA probes 16 .<br />
61