Compendium of Potato Diseases - (PDF, 101 mb) - USAID
Compendium of Potato Diseases - (PDF, 101 mb) - USAID
Compendium of Potato Diseases - (PDF, 101 mb) - USAID
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
WAI.KINSHtAW, C. ].. and R.11.LARSON. 1959. Corky ringspot <strong>of</strong><br />
potato. A soil-borne virus disease. Wis. Agric. Exp. Sin. Res. Bull.<br />
217. 31 pp.<br />
(Prepared by 1). P. Weingartner)<br />
<strong>Potato</strong> Yellow Dwarf Virus<br />
<strong>Potato</strong> yellow dwarf virus (PYDV) occurs in Canada and in<br />
the United States in Michigan, New York. and Wisconsin.<br />
Disease outbreaks have not been reported for almost 40 years.<br />
Symptoms<br />
Vines from infected seed pieces are dwarfed and brittle, and<br />
Vine eedpiees frm ifeced re wared nd ritleand<br />
the entire plant has a yellowish cast. I.eaflet margins roll<br />
upward, but the longitudinal axis <strong>of</strong> the leaflet curves<br />
downward (Plate 71). Pith necrosis <strong>of</strong> stems is common,<br />
appearing shortly after foliage chlorosis. Necrosis beginning<br />
near the growing point may eventually extend the length <strong>of</strong> the<br />
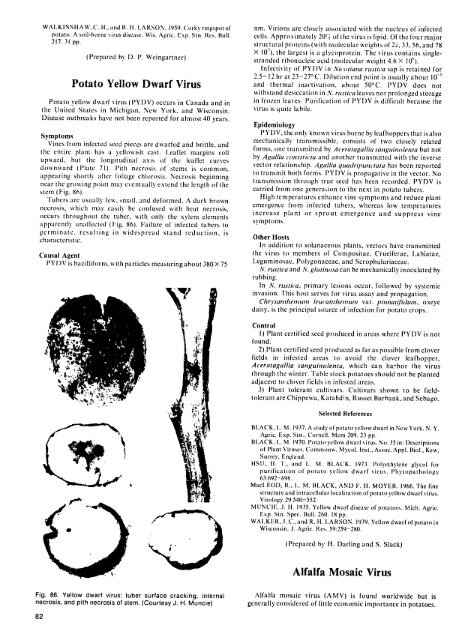
stem (Fig. 86).<br />
Tubcrs are usulally few, small, and deformed. A dark brown<br />
necrosis, which may easily he confused with heat necrosis,<br />
occurs<br />
i<br />
throughout the tuber, with only the xylem elements<br />
apparently unaffected (Fig. 86). Failure <strong>of</strong> infected tubers to<br />
germinate, resulting in widespread stand reduction, is<br />
characteristic.<br />
Causal Agent<br />
'YI)V isbacilliform, with particles measuring about 380 X 75<br />
It.<br />
*;<br />
fVirology<br />
Fig. 86. Yellow dwarf virus: tuber surface cracking, internal<br />
necrosis, and pith necrosis <strong>of</strong> stem. (Courtesy J. H. Muncie)<br />
82<br />
nm. Virions are closely associated with the nucleus <strong>of</strong> infected<br />
cells. Approximately 20%' <strong>of</strong> the virus is lipid. Of the four major<br />
structural proteins (with molecular weights <strong>of</strong> 22, 33, 56, and 78<br />
X 10 ). the largest is a glycoprotein. The virus contains singlestranded<br />
ribonucleic acid (molecular weight 4.6 X 10"').<br />
Infectivity <strong>of</strong> PYI)V in Nicotiana rustica sap is retained for<br />
2.5-12 hr at 23-27 -<br />
0 C. Dilution end point is usually about 10<br />
and thermal inactivation, about 50'C. IPYDV does not<br />
withstand desiccation in N. rustica leaves nor prolonged storage<br />
in frozen leaves. Purification <strong>of</strong> PYI)V is difficult because the<br />
virus is quite labile.<br />
Epidemiology<br />
PYDV, the only known virus borne by leafhoppers that isalso<br />
mechanically transmissible, consists <strong>of</strong> two closely related<br />
forms, one transmitted by Aceratagallia sanguinolenita but not<br />
byrAgalona<br />
by Agallia<br />
transmittedand another transmitted wit t nr<br />
constrictaand another tiansmitted with tile inverse<br />
vector relationship. Agalha quadrupuncata has been reported<br />
to transmit both forms. PYDV is propagative in the vector. No<br />
transmission through true seed his been recorded. PYDV is<br />
c r m o erion t oee xt in poaor .<br />
carried from one generation to the next in <strong>Potato</strong> tubers.<br />
High temperatures enhance vine symptoms and reduce plant<br />
emergence from infected tubers, whereas low temperatures<br />
increase plant or sprout emergence and suppress vine<br />
s p l s mu s<br />
symptoms.<br />
Other Hosts<br />
In addition to solanaceous plants, vectors have transmitted<br />
the virus to me<strong>mb</strong>ers <strong>of</strong> Compositac, Cruciferae, Labiatae,<br />
Leguminosae. Polygonaceae, and Scrophulariaccae.<br />
N. rushica and N.glutinosa can be mechanically inoculated by<br />
rubbing.<br />
In N. rustica, primary lesions Occur, followed by systemic<br />
invasion. This host serves for virus assay and propagation.<br />
hChr.santhemtnn leucanthetan var. pinnatqiidn, oxeye<br />
daisy. is the principal source <strong>of</strong> infection for potato crops.<br />
Control<br />
I) Plant certified seed produced in areas where PYDV is not<br />
found.<br />
2) Plant certified seed produced as far as possible from clover<br />
fields in infested areas to avoid the clover leafhopper,<br />
Aceratagallia sanguinolenta, which can harbor the virus<br />
through the winter. Table stock potatoes should not be planted<br />
adjacent to clover fields in infested areas.<br />
3) Plant tolerant cultivars. Cultivars shown to be fieldtolerant<br />
are Chippewa, Katahdin. Russet Burbank, and Sebago.<br />
Selected References<br />
BI.ACK, I.. M. 1937. A study l<strong>of</strong>'potato yellowdwarf in New York. N.Y.<br />
Agric. Exp. Sin.. Cornell. Mem 209. 23 pp.<br />
BLACK. I..M. 1970. <strong>Potato</strong> yellow dwarf virus. No. 35 in: Descriptions<br />
<strong>of</strong> Plant Viruses. Cormonw,. Mycol. Inst., Assoc. Appl. Biol., Kew,<br />
Surrey. England.<br />
HStU, II. T.. and .. M. BI.ACK. 1973. Polyethylene glycol for<br />
purification <strong>of</strong> potato yellow dwarf virus. Phytopathology<br />
63:692-696.<br />
MacI.EOI), R., . M. BL+ACK, AND F. H. MOYER. 1966. The fine<br />
structure and intracellular localiation <strong>of</strong> potato yellow dwarf virus.<br />
29:540-552.<br />
tMUNCIE, .1.II. 1935. Yellow dwarf disease <strong>of</strong> potatoes. Mich. Agric.<br />
Exp. Stn. Spec. Bull. 260. 18 pp.<br />
WAI.KER,-1. C.. and R. II. I.ARSON. 1939. Yellowdwarf <strong>of</strong>potato in<br />
Wisconsin. .1.Agric. Res. 59:259-280.<br />
(Prepared by H. Darling and S. Slack)<br />
Alfalfa Mosaic Virus<br />
Alfalfa mosaic virus (AMV) is found worldwide but is<br />
generally considered <strong>of</strong> little economic importance in potatoes.