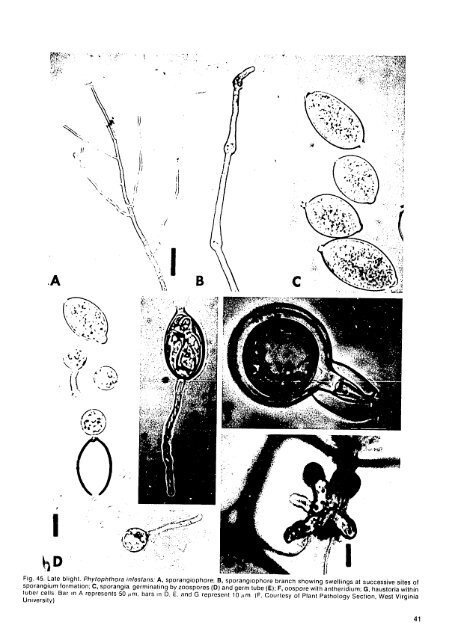
Disease Cycle P. er t/lro.sptica is soilhorne and endemic in many soils, Zoospores, sporangia, or oospores may serve as inoculum, but oospores are probably the significant propagule in pathogen dissemination and survival in soil. Plants <strong>of</strong> all ages are susceptible, but the disease is most frequently observed in mature plants approaching harvest, Epidemiology Disease develops in soils approaching saturation from poor drainage or excessive precipitation or irrigation. l.arge amounts <strong>of</strong>decomposing plant residues in soil enhance waterabsorption, retention, and disease incidence In wet soil, the disease develops overa wide range <strong>of</strong>temperatures but is most severe between 20 and 30' C. Other Hosts Although I'. erth'irosepti'ahas been reported as a pathogen only on potatoes and tulips, it has been recovered from the roots <strong>of</strong> 17 nonsolanaceous plants, including wheat and rye. Control I) Plant seed tubers in soils with good drainage, 2) Avoid excessive irrigation late in the growing season. Selected References GOSS. R. W. 1949. i Inyk rot <strong>of</strong> poaioes caused by Hll.1i.tfora £'rtroWIi- 1thN . Neh. Agric. T Fxp. Sin. Re. Bull. 160. 27 ROWE, E.C.. and A.F.SCHlMIlTlHlNNlER. 1977. <strong>Potato</strong> pink rot in Ohio caused hy /'htropilthora ertihro. i/c'T 1(In I'. rrptogea. plant Dis. Rep. 61:80t7-81t0. STAMPS, 1).J. 1978. 'hoitop/hihoraerrth-.7pti(a. No. 593 in: t)escriptions <strong>of</strong> IPaihogenic hungi and Bacteria. C'mmonw. Mycol. Inst.. Kew. Surrey, l-ngland. 2 pp. VARGAS, I.. A.. and .. W. NIELSEN. 1972. I'hvtoph/hora rithro.T'apOtt. ini'Peru: Its identilication and pathogenesis. Am. WAtI.R IOUSE. . 1963. Key to the species <strong>of</strong> I'hthra Ie WarS. No. 92 in: M'co. Kipers ommo,,. Mycol. Inst.. Kew. Surre ' Eingland. 22 pp. (Prepared by R. C. Rowe and L. W. Nielsen) Late Blight Late blight is probably the single most important disease <strong>of</strong> potatoes worldwide. It is destructive wherever potatoes are grown without fungicides, except in hot, dry, irrigated areas. The Irish potato famine" <strong>of</strong> the 1840s was caused by PIeqopititorainfisians, the fungus that causes late blight. Immense quantities <strong>of</strong> fungicides are applied to potatoes throughout the world for protection against P. inf'sian. Symptoms moisture, Leaf lesions light are intensity, highly and variable, host cultivar. depending Initial on smptomsare temperature, typically small, pale to dark green, irregularly shaped spots. Under favorable environmental conditions they rapidly grow to large, brown to purplish black, necrotic lesions that -ay kill entire leaflets and spread via petioles to the stem, ev, .itually killing the entire plant. A pale green to yel!ow halo is <strong>of</strong>ten present outside the area <strong>of</strong> leaf necrosis (Plate 28). Under moist conditions, a white downy mildew <strong>of</strong> sporangia and sporangiophores appears at the edge <strong>of</strong> lesions, mostly on the underside <strong>of</strong> the leaves (Plate 29). In the field, plants severely affected with late blight give <strong>of</strong>fa distinctive odor. This odor actually results from rapid breakdown <strong>of</strong> potato leaf tissue and also follows chemical vine killing, frosts, etc. Positive identification <strong>of</strong> late blight requires confirmation <strong>of</strong> sporangia and sporangiophores either on lesions in the field 40 under moist conditions or on leaf or tuber lesions incubated in a moist cha<strong>mb</strong>er. On susceptible cultivars, exteriors <strong>of</strong> infected tubers show irregular, small to large, slightly depressed areas <strong>of</strong> brown to purplish skin (Plate 30). A tan-brown, dry granular rot characteristically extends into the tuber approximately 1.5 cm, the depth varying according to length <strong>of</strong> time after infection, cultivar, and temperature. The boundary between diseased and healthy tissue is not clearly defined; delicate, brown, peglike extensions penetrate to variable depths. Under cool, dry storage conditions, tuber lesions develop slowly and may become slightly sunken after several months. Secondary organisms (bacteria and fungi) <strong>of</strong>ten follow infection by '. it/i,stans, resulting in partial or complete breakdown <strong>of</strong> tubers and complicating diagnosis. Causal Organism Phytoplithora infi 'sans(Mont.) de lary has sporangia (conidia) that are hyaline, lemon-shaped, thin-walled, and 21-38 X 12-23 mm in size. Each has an apical papillum (Fig. 45C). Sporangia <strong>of</strong> P. infestan.s are borne on the tip <strong>of</strong> a sporangiophore branch (Fig. 45A); as it elongates, the sporangiophore swells slightly and turns the attached sporangium to the side. The sporangiopore is thus characterized by periodic swellings (Fig. 4513) at points where sporangia were produced. Sporangia may germinate by means <strong>of</strong> a germ tube (Fig. 45E), but most commonly they form about eight biciliate zoospores (Fig. 45D) that swim freely in waterand encyst on solid surfaces. i'ncysted zoospores can germinate by germ tubes that enter thhotvalfsomabtualyanpreoiu isomd the host via leaf stomata, but usuallyanappressoriumisformed and penetration hyphae enter directly through the cuticle. Once inside the plant, the nonseptate mycelium is intercellular and intracellular by means <strong>of</strong> haustoria (Fig. 45G) that extend into cells. Sexual reproduction resultsin oospores(Fig. 45F) formed by the union <strong>of</strong> oogonia and antheridia. Oospores within oogonia are 24 -46 pm in diameter and germinate via a germ tube with a terminal sporangium, which, in turn, either liberates zoospores or forms another germ tube. Disease Cycle Oospores in nature have been found only in Mexico, where both mating types (A 1 and A.) occur. Leaves touching the soil are <strong>of</strong>ten infected first, suggesting that oospores probably playa role in the survival <strong>of</strong> P.itnrestarisunder adverse conditions. In tropical areas where the crop is grown all year, overwintering <strong>of</strong> R itirestars is not an important consideration. However, where distinct seasons occur, P. ittf'stans overwinters as on mycelium farms or in near unharvested commercial tubers, storages, tubers or dumped tubers in stored cull piles saved for seed. After plant emergence, the fungus and invades a few <strong>of</strong> the growing sprouts and sporulates under moist conditions, producing primary inoculum. Once primary infection has occured, further spread <strong>of</strong> P. inl.san. takes place by airborne or waterborne sporangia (Fig. 46). <strong>of</strong> Tubers succulent on tissue cull piles that frequently are easily infected sprout and by form . it/.Vllns dense masses frotm diseased tubers. spores Spoi ulation within the foliage mass produces pr digious nu<strong>mb</strong>ers f sp res to infect nearby fields. Epidemiology Tubers, particularly those inadequately covered by soil, may be infected in the field by spores that have been washed from infected leaves into the soil by rain or irrigation. Rapid tuber growth frequently causes soil to crack, exposing tubers to infection. Tuber infection may also occur during wet harvest conditions via contact between ttibers and sporangia on vines or via airborne sporangia. Little, if any, spread <strong>of</strong> P itiA,stans occurs under optimum conditions in storage. Field infection is most successful under cool, moist conditions. lowever, infections take place over a range <strong>of</strong> environmental conditions, and high temperature strains <strong>of</strong> the
'40 ff .. . '1 f . ."L', . '1 .,. " . .. ;, B C .. :-,I -, .141 Fig. 45. Late blight. Phytophthora Infostans: A, sporangiophore; B, sporangiophore branch showing swellings at successive sites <strong>of</strong> sporangium formation; C, sporangia, germinating by zoospores (D) and germ tube (E=);F, oospore with antheridium; G, haustoria within tuber cells Bar in A represents 50 pm. hars in D. E. and G represent 10 pm. (F. Courtesy <strong>of</strong> Plant Pathology Section, West Virginia University) 41
- Page 1 and 2: , ,. ,o . ; , . o , . r. , -' .L ,
- Page 3 and 4: Compendium of Potato Diseases W. J.
- Page 5 and 6: In memory ofimy respected colleague
- Page 7 and 8: Acknowledgments Planning Committee
- Page 9 and 10: Introduction 1 Potato Disease 1 The
- Page 11 and 12: A potato disease is an interaction
- Page 13 and 14: Fig. 1.A, Lower portion of young po
- Page 15 and 16: +q I B Fig 4. Natural openings in a
- Page 17 and 18: Part I. Disease in the Absence of I
- Page 19 and 20: more heavily in the vascular region
- Page 21 and 22: the air temperature when tubers are
- Page 23 and 24: iiO A B 'D C Fiq. 14 Second growth:
- Page 25 and 26: identifying tissue bruised by black
- Page 27 and 28: Internal Sprouting multiple sprouts
- Page 29 and 30: conditions permits normal leaf deve
- Page 31 and 32: Epidemiology Oxidant injury is pres
- Page 33 and 34: sevritof stnting, chiorosis. loss o
- Page 35 and 36: Selected Reference brown to bronze
- Page 37 and 38: Part II. Disease in the Presence of
- Page 39 and 40: should be planted on land with at l
- Page 41 and 42: Control I) Use disease-free tubers
- Page 43 and 44: FOI-SO M. D, and I. A. FRIEDMAN. )9
- Page 45 and 46: Fungi Powdery Scab liberating powde
- Page 47 and 48: Histopathology Sort of sporangia de
- Page 49: any time but generally occurs late
- Page 53 and 54: A \, 2) Powdery mildew is rarely a
- Page 55 and 56: R., Inst.. Kew. Surrey. ngland. 609
- Page 57 and 58: TORRES, H.. E. R. FRENCH, and I.. W
- Page 59 and 60: d AC D A 0 Fig 53 Gray mold. A, Bot
- Page 61 and 62: strands of pigmented hyphae. Sclero
- Page 63 and 64: tuber malformation. Roots are also
- Page 65 and 66: 1. Giant-hill plants, taller than n
- Page 67 and 68: 13. Bacterial soft rot. Erwinia car
- Page 69 and 70: 24. Wart. Synchytrium endobioticum
- Page 71 and 72: 34. Pleospora herbarum (Stemphylium
- Page 73 and 74: 44. Charcoal rot. Macrophomina phas
- Page 75 and 76: 55. Leafroll virus. Current season
- Page 77 and 78: 67. Mop-top virus. Primary tuber 68
- Page 79 and 80: 77. Aster yellows mycoplasma sympto
- Page 81 and 82: Symptoms Small, localized, light br
- Page 83 and 84: obovoid, and 14-30 X 5-10 pm. Pycni
- Page 85 and 86: invaders through the IFusarium lesi
- Page 87 and 88: necrosis extending into the tuber t
- Page 89 and 90: do not survive drying. Both species
- Page 91 and 92: cauisal. Tesis Magister Scientiae e
- Page 93 and 94: penetration, and subsequent disease
- Page 95 and 96: have been demonstrated, including t
- Page 97 and 98: In P..floridana,strains of PVY" and
- Page 99 and 100: infects certain comnie,-cial stocks
- Page 101 and 102:
cytoplasm of infected potato cells
- Page 103 and 104:
WETTER, C. 1971. Potato virus S. No
- Page 105 and 106:
3) Resistance has not been identifi
- Page 107 and 108:
nm), which do code for coat protein
- Page 109 and 110:
Symptoms AMV may induce Other predo
- Page 111 and 112:
Disease Cycle I)escriptions of Plan
- Page 113 and 114:
Selected References KASSANIS, B. 19
- Page 115 and 116:
Control 1)Avoid locations in wkhich
- Page 117 and 118:
The "yellows" types of disease, cha
- Page 119 and 120:
modifying cultural practices and by
- Page 121 and 122:
immature females in the white or ye
- Page 123 and 124:
l.aboratory, Control. and Quarantin
- Page 125 and 126:
They attack many major crops in the
- Page 127 and 128:
infested areas of North America sev
- Page 129 and 130:
antennal tb l tuberce u antenna eye
- Page 131 and 132:
carbon tetrachloride, applied at th
- Page 133 and 134:
Diagnostic Microbial Structures Scl
- Page 135 and 136:
Surface and or interior Shades of g
- Page 137 and 138:
Equivalent Names of Potato Diseases
- Page 139 and 140:
Common Name Causal Factor Other Nam
- Page 141 and 142:
Common Name Causal Factor Other Nam
- Page 143 and 144:
coalesce-union of similar structure
- Page 145 and 146:
many days following removal of the
- Page 147 and 148:
Index Abrasions, tuber surfaces. 14
- Page 149:
Irost tolerance in. 9 aniligena. 68