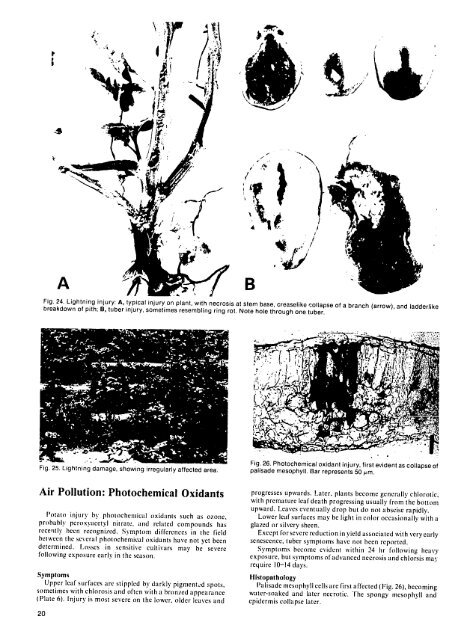
IAA 94 / I Am /N F. n iFig. 26. Photochemical oxidant injury, first evident as collapse <strong>of</strong> Fig. 25. Lightning damage, showing irregularly affected area. palisade mesophyll. Bar represents 50 pm. Air Pollution: Photochemical Oxidants progresses upwards. Later, plants become generally chlorotic, with premature leaf death progressing usually from the bottom upward. <strong>Potato</strong> Leaves eventually injury by photochemical drop but do not oxidants abscise rapidly. such as ozone, Lower leaf surfaces may be light in color occasionally with a probably peroxyacetyl nitrate, and related compounds has glazed or silvery sheen. recently been recognized. Symptom differences in the field Except for severe reduction in yield associated between with the veryearly several photochemical oxidants have not yet been senescence, tuber symptoms have not been reported. determined. Losses in sensitive cultivars may be severe Symptoms become evident within 24 hr following following heavy exposure early in the season, exposure, but symptoms <strong>of</strong> advanced necrosis and chlorsis may require 10-14 days. Symptoms Histopathology Upper leaf surfaces are stippled by darkly pigmented spots, Palisade mesophyll cells are first affected (Fig. 26), becoming sometimes with chlorosis and <strong>of</strong>ten with a bronzed appearance water-soaked and later necrotic. The spongy mesophyll and (Plate 6). Injury is most severe on the lower, older leaves and epidermis collapse later. 20
Epidemiology Oxidant injury is present in North America along the Atlantic coast, in the Great Lakes region, the southeastern states, and the Pacific southwest. Photoche-:cal oxidants accumulate under two conditions: when relati, - large areas <strong>of</strong> high atmospheric pressure are present or wl.en air masses stagnate under a layer <strong>of</strong> warm air over cool land surfaces. Episodes in which normal dispersion <strong>of</strong> air pollutants is prevented may occur infrequently during the growing season. Extent <strong>of</strong> injury is influenced by the concentration <strong>of</strong> oxidants, length and frequency <strong>of</strong> exposure, plant genotypc, and stage <strong>of</strong> plant growth. Field exposures <strong>of</strong> approximately 0.15 ppm ozone for a day or two are usually sufficient to injure xposed foliage. The amount <strong>of</strong>daniage islargely influenced by density <strong>of</strong> the foliage mass. If the foliage mass is sufficiently large, a "sink" effect is produced, by which air pollutants are absorbed or adsorbed by leaf surfaces and removed from the immediate environment, thereby protecting nearby foliage. Thus, exposed leaves above the foliage canopy may he severely damaged and leaves within the canopy escape injury. Injury may be more severe at field margins than in the center. If plants are small, leaf and stem exposure is complete and the sink effect is negligible; thus plant injury may be severe following an earl) season episode. Ozone injury can predispose potato leaves to Boir tiscinerea infection and may increase susceptibility to other pathogens. Control Wide varietal differences in tolerance exist. Cultural practices stimulating vigorous early season vine growth may hasten plants past the susceptible, small vine stage. Maintenance <strong>of</strong> a heavy foliage canopy until the tuber crop is assured may lessen or avoid midseason injury, Selectd References BRASIIER, E.P.,I). J. FIELI)HOUSE, and M. SASSER. 1973. (/one injury in potato variety trials. Plant l)is. Rep. 57:542-544. HEGGESTAD, H. E. 1973. Photochemical air pollution injury to potatoes in the Atlantic Coastal States. Am. <strong>Potato</strong> J. 50: 315-328. HOOKER. W.., T. C. YANG, and H. S. POTTER. 1973. Air pollution injurv <strong>of</strong> potato in Michigan. Am. <strong>Potato</strong> J. 50:151-161. MANNING, W. J..W. A. FEDER. 1.PERKINS, and M. GLICK MAN. 1969. Oione injury and infection <strong>of</strong> potato leaves by Botrviis ciwerea. Plant Dis. Rep. 53:691-693. MOSI.tY. A. R., R. C. ROWE, and T.C. WEIDENSAUL. 1978. Relationship <strong>of</strong> foliar ozone injury to maturity classification and yield <strong>of</strong> potatoes. Am. <strong>Potato</strong> .1.55:147-153. (Prepared by W. J. Hooker) Air Pollution: Sulfur Oxides Although potato leaves are relatively resistant to injury by sulfur oxides, they re-pond with iaiterveinal necrotic areas that are light tan to white (Plate 7), and yields may be reduced. Injury should be anticipated in areas with air flow drainage patterns downwind from power plants and smelters. If sulfur oxides a-r injuring potatoes, symptoms on nearby sensitive plants (alfalfa, bean, soybean, beet. /1maranthus spp., bindweed, morning glory, lettuce, curly dock, plantain, ragweed, or sunflower) should confirm the diagnosis. Selected References JONES, H. C.. 1). WEBER, and 1). BALSILI.IE. 1974. Acceptable limits for air pollution dosages and vegetation effects: Sulfur dioxide. Paper No. 74-225. Air Pollution Control Assoc. 67th Annual Meeting, )enver. THOMAS, M. I)., and R. H. HENDRICKS. 1956. Effect <strong>of</strong> air pollution on plants. Section 9. pages 1-44 in: P. I.. Magill, F. R. Holden. and C.Ackley. eds. Air Pollution Handbook. McGraw- Hill. New York. (Prepared by W. J. Hooker) Chemical Injury A wide range <strong>of</strong> chemicals accidentally or improperly applied can cause divergent symptoms on foliage and in tubers, with severity depending upon the nature <strong>of</strong> the chemical, its dosage, environmental factors, and plant maturity and variety. Vinekilling preharvest defoliants frequently cause necrosis at the stolon attachment and vascular discoloration <strong>of</strong> the stem end, rese<strong>mb</strong>ling symptoms <strong>of</strong> stem-end browning or Verticillium wilt. Interveinal leaf tissues may be burned. Moisture stress increases symptom severity. Growth-regulating herbicides for weed control in potatoes or herbicide-s airborne from nearby areas may cause leaf distortion superficially suggesting virus infection (Fig. 27A and B). Tuber skin color may be affected. Some ( 2 ,4,5-trichlorophenoxyacetates) cause necrosis not unlike that from severe deep scab and also tuber deformation (Figs. 27C and D). In storage, netting <strong>of</strong> tuber surfaces and dehydration have followed foliage application <strong>of</strong> maleic hydrazide; abnormal sprouting has been associated with other compounds. (See internal sprouting.) Improper application <strong>of</strong> fertilizer to foliage or application too close to the seed piece in the soil causes foliage or seed tuber necrosis, followed by decay, poor stands, and low plant vigor. Selected References FRYER, J. D., and R.J. MAKEPEACE, eds. 1972. Weed Control Handbook. Vol. 2. Recommendations Including Plant Growth Regulations, 7th ed. Blackwell Scientific Publications, London. 424 HOOKER, W. J., and A. F. SHERF. 1951. Scab susceptibility and injury <strong>of</strong> potato tubers by 2 ,4,5-trichlorophenoxyacetates. Am. <strong>Potato</strong> J. 28:675-681. MUNSTER, J.,and P. CORNU. 1971. D~gts interne3 causes aux tubercules de pommes de terre par lasfcheresse ou par I'application de reglone. Rev. Suisse Agric. 3:55-59. MURPHY, H. J. 1968. <strong>Potato</strong> vine killing. Am. <strong>Potato</strong> J. 45:472-478. POABST, P. A., and C. GENIER. 1970. A storage disorder in D Fig. 27. Chemical injury: A, burn <strong>of</strong> interveinal leaf tissue; B, Leaf deformation by growth-regulating herbicide; C and D, tuber injury from foliaga application <strong>of</strong> 2,4,5-trichlorophenoxyacetate. (C and D,Courtesy W.J. Hooker and A. F. Sherf) 21
- Page 1 and 2: , ,. ,o . ; , . o , . r. , -' .L ,
- Page 3 and 4: Compendium of Potato Diseases W. J.
- Page 5 and 6: In memory ofimy respected colleague
- Page 7 and 8: Acknowledgments Planning Committee
- Page 9 and 10: Introduction 1 Potato Disease 1 The
- Page 11 and 12: A potato disease is an interaction
- Page 13 and 14: Fig. 1.A, Lower portion of young po
- Page 15 and 16: +q I B Fig 4. Natural openings in a
- Page 17 and 18: Part I. Disease in the Absence of I
- Page 19 and 20: more heavily in the vascular region
- Page 21 and 22: the air temperature when tubers are
- Page 23 and 24: iiO A B 'D C Fiq. 14 Second growth:
- Page 25 and 26: identifying tissue bruised by black
- Page 27 and 28: Internal Sprouting multiple sprouts
- Page 29: conditions permits normal leaf deve
- Page 33 and 34: sevritof stnting, chiorosis. loss o
- Page 35 and 36: Selected Reference brown to bronze
- Page 37 and 38: Part II. Disease in the Presence of
- Page 39 and 40: should be planted on land with at l
- Page 41 and 42: Control I) Use disease-free tubers
- Page 43 and 44: FOI-SO M. D, and I. A. FRIEDMAN. )9
- Page 45 and 46: Fungi Powdery Scab liberating powde
- Page 47 and 48: Histopathology Sort of sporangia de
- Page 49 and 50: any time but generally occurs late
- Page 51 and 52: '40 ff .. . '1 f . ."L', . '1 .,. "
- Page 53 and 54: A \, 2) Powdery mildew is rarely a
- Page 55 and 56: R., Inst.. Kew. Surrey. ngland. 609
- Page 57 and 58: TORRES, H.. E. R. FRENCH, and I.. W
- Page 59 and 60: d AC D A 0 Fig 53 Gray mold. A, Bot
- Page 61 and 62: strands of pigmented hyphae. Sclero
- Page 63 and 64: tuber malformation. Roots are also
- Page 65 and 66: 1. Giant-hill plants, taller than n
- Page 67 and 68: 13. Bacterial soft rot. Erwinia car
- Page 69 and 70: 24. Wart. Synchytrium endobioticum
- Page 71 and 72: 34. Pleospora herbarum (Stemphylium
- Page 73 and 74: 44. Charcoal rot. Macrophomina phas
- Page 75 and 76: 55. Leafroll virus. Current season
- Page 77 and 78: 67. Mop-top virus. Primary tuber 68
- Page 79 and 80: 77. Aster yellows mycoplasma sympto
- Page 81 and 82:
Symptoms Small, localized, light br
- Page 83 and 84:
obovoid, and 14-30 X 5-10 pm. Pycni
- Page 85 and 86:
invaders through the IFusarium lesi
- Page 87 and 88:
necrosis extending into the tuber t
- Page 89 and 90:
do not survive drying. Both species
- Page 91 and 92:
cauisal. Tesis Magister Scientiae e
- Page 93 and 94:
penetration, and subsequent disease
- Page 95 and 96:
have been demonstrated, including t
- Page 97 and 98:
In P..floridana,strains of PVY" and
- Page 99 and 100:
infects certain comnie,-cial stocks
- Page 101 and 102:
cytoplasm of infected potato cells
- Page 103 and 104:
WETTER, C. 1971. Potato virus S. No
- Page 105 and 106:
3) Resistance has not been identifi
- Page 107 and 108:
nm), which do code for coat protein
- Page 109 and 110:
Symptoms AMV may induce Other predo
- Page 111 and 112:
Disease Cycle I)escriptions of Plan
- Page 113 and 114:
Selected References KASSANIS, B. 19
- Page 115 and 116:
Control 1)Avoid locations in wkhich
- Page 117 and 118:
The "yellows" types of disease, cha
- Page 119 and 120:
modifying cultural practices and by
- Page 121 and 122:
immature females in the white or ye
- Page 123 and 124:
l.aboratory, Control. and Quarantin
- Page 125 and 126:
They attack many major crops in the
- Page 127 and 128:
infested areas of North America sev
- Page 129 and 130:
antennal tb l tuberce u antenna eye
- Page 131 and 132:
carbon tetrachloride, applied at th
- Page 133 and 134:
Diagnostic Microbial Structures Scl
- Page 135 and 136:
Surface and or interior Shades of g
- Page 137 and 138:
Equivalent Names of Potato Diseases
- Page 139 and 140:
Common Name Causal Factor Other Nam
- Page 141 and 142:
Common Name Causal Factor Other Nam
- Page 143 and 144:
coalesce-union of similar structure
- Page 145 and 146:
many days following removal of the
- Page 147 and 148:
Index Abrasions, tuber surfaces. 14
- Page 149:
Irost tolerance in. 9 aniligena. 68