Biomedical Research in Developing Countries - UNICRI
Biomedical Research in Developing Countries - UNICRI
Biomedical Research in Developing Countries - UNICRI
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
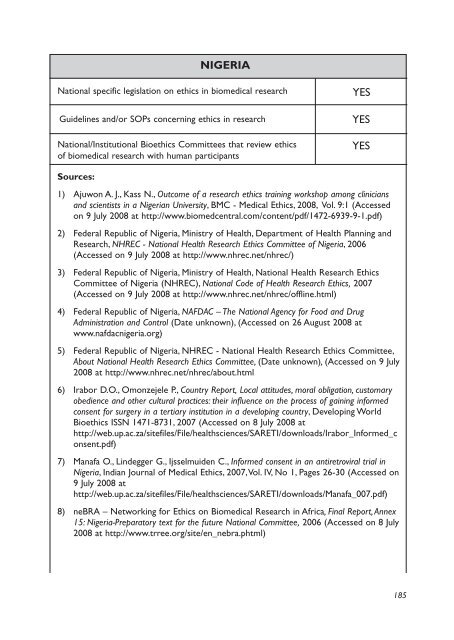
NIGERIA<br />
National specific legislation on ethics <strong>in</strong> biomedical research<br />
Guidel<strong>in</strong>es and/or SOPs concern<strong>in</strong>g ethics <strong>in</strong> research<br />
National/Institutional Bioethics Committees that review ethics<br />
of biomedical research with human participants<br />
YES<br />
YES<br />
YES<br />
Sources:<br />
1) Ajuwon A. J., Kass N., Outcome of a research ethics tra<strong>in</strong><strong>in</strong>g workshop among cl<strong>in</strong>icians<br />
and scientists <strong>in</strong> a Nigerian University, BMC - Medical Ethics, 2008, Vol. 9:1 (Accessed<br />
on 9 July 2008 at http://www.biomedcentral.com/content/pdf/1472-6939-9-1.pdf)<br />
2) Federal Republic of Nigeria, M<strong>in</strong>istry of Health, Department of Health Plann<strong>in</strong>g and<br />
<strong>Research</strong>, NHREC - National Health <strong>Research</strong> Ethics Committee of Nigeria, 2006<br />
(Accessed on 9 July 2008 at http://www.nhrec.net/nhrec/)<br />
3) Federal Republic of Nigeria, M<strong>in</strong>istry of Health, National Health <strong>Research</strong> Ethics<br />
Committee of Nigeria (NHREC), National Code of Health <strong>Research</strong> Ethics, 2007<br />
(Accessed on 9 July 2008 at http://www.nhrec.net/nhrec/offl<strong>in</strong>e.html)<br />
4) Federal Republic of Nigeria, NAFDAC – The National Agency for Food and Drug<br />
Adm<strong>in</strong>istration and Control (Date unknown), (Accessed on 26 August 2008 at<br />
www.nafdacnigeria.org)<br />
5) Federal Republic of Nigeria, NHREC - National Health <strong>Research</strong> Ethics Committee,<br />
About National Health <strong>Research</strong> Ethics Committee, (Date unknown), (Accessed on 9 July<br />
2008 at http://www.nhrec.net/nhrec/about.html<br />
6) Irabor D.O., Omonzejele P., Country Report, Local attitudes, moral obligation, customary<br />
obedience and other cultural practices: their <strong>in</strong>fluence on the process of ga<strong>in</strong><strong>in</strong>g <strong>in</strong>formed<br />
consent for surgery <strong>in</strong> a tertiary <strong>in</strong>stitution <strong>in</strong> a develop<strong>in</strong>g country, Develop<strong>in</strong>g World<br />
Bioethics ISSN 1471-8731, 2007 (Accessed on 8 July 2008 at<br />
http://web.up.ac.za/sitefiles/File/healthsciences/SARETI/downloads/Irabor_Informed_c<br />
onsent.pdf)<br />
7) Manafa O., L<strong>in</strong>degger G., Ijsselmuiden C., Informed consent <strong>in</strong> an antiretroviral trial <strong>in</strong><br />
Nigeria, Indian Journal of Medical Ethics, 2007,Vol. IV, No 1, Pages 26-30 (Accessed on<br />
9 July 2008 at<br />
http://web.up.ac.za/sitefiles/File/healthsciences/SARETI/downloads/Manafa_007.pdf)<br />
8) neBRA – Network<strong>in</strong>g for Ethics on <strong>Biomedical</strong> <strong>Research</strong> <strong>in</strong> Africa, F<strong>in</strong>al Report, Annex<br />
15: Nigeria-Preparatory text for the future National Committee, 2006 (Accessed on 8 July<br />
2008 at http://www.trree.org/site/en_nebra.phtml)<br />
185