- Page 1 and 2: Aspen Physical Property System Phys
- Page 3 and 4: Contents Contents..................
- Page 5 and 6: General Pure Component Solid Molar
- Page 7 and 8: 1 Introduction This manual describe
- Page 9 and 10: Pure Component Temperature- Depende
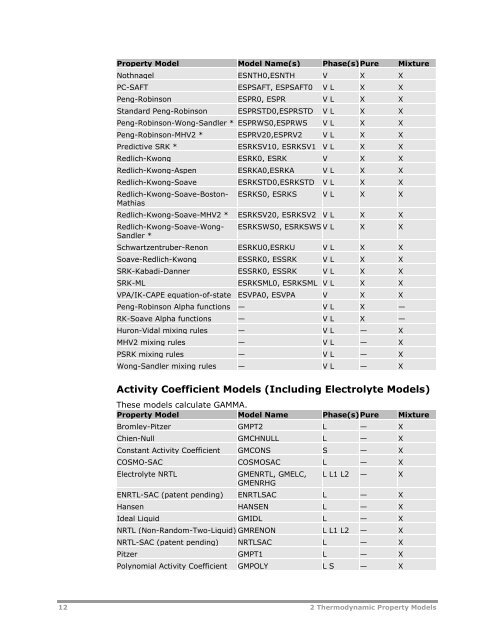
- Page 11 and 12: the Properties Parameters Pure-Comp
- Page 13: 2 Thermodynamic Property Models Thi
- Page 17 and 18: Property Model Model Name 2 Thermod
- Page 19 and 20: Where: Parameter Name/Element 2 The
- Page 21 and 22: critical temperature, the critical
- Page 23 and 24: Where the ideal-gas contribution
- Page 25 and 26: , where � For polar, associating
- Page 27 and 28: Parameter Name/ Element 2 Thermodyn
- Page 29 and 30: The denominator of equation 8 is gi
- Page 31 and 32: V. V. De Leeuw and S. Watanasiri, "
- Page 33 and 34: References M. Benedict, G. B. Webb,
- Page 35 and 36: References L. Haar, J.S. Gallagher,
- Page 37 and 38: Parameter Name/Element NTHDDH 0 †
- Page 39 and 40: Gibbs free energy departure: The fo
- Page 41 and 42: For a block copolymer, there is onl
- Page 43 and 44: Copolymer PC-SAFT Dispersion Term T
- Page 45 and 46: The association-site number of the
- Page 47 and 48: Then I2(�) and I3(�) are comput
- Page 49 and 50: A databank called PC-SAFT contains
- Page 51 and 52: Parameter Name/Element PRKBV/1 k ij
- Page 53 and 54: Predictive SRK (PSRK) This model us
- Page 55 and 56: ka,ij kb,ij = = For best results, b
- Page 57 and 58: J. Schwartzentruber and H. Renon, "
- Page 59 and 60: Redlich-Kwong-Soave-MHV2 This equat
- Page 61 and 62: ‡ RKUC0, RKUC1, and RKUC2 are tre
- Page 63 and 64: Parameter Name/ Element SRKLIJ/3 l
- Page 65 and 66:
Parameter Name/ Element Symbol Defa
- Page 67 and 68:
Association Equilibria Every associ
- Page 69 and 70:
and respectively. For the reactions
- Page 71 and 72:
R. W. Long, J. H. Hildebrand, and W
- Page 73 and 74:
Where the L, M, and N are parameter
- Page 75 and 76:
Alpha function Model name First Opt
- Page 77 and 78:
mi = Computed by equation 6 Equatio
- Page 79 and 80:
Parameter Name/Element 2 Thermodyna
- Page 81 and 82:
standard RKS alpha function, except
- Page 83 and 84:
These mixing rules are discussed se
- Page 85 and 86:
Where both Ai* and Am are calculate
- Page 87 and 88:
Like Huron and Vidal, the limiting
- Page 89 and 90:
Model Type Wilson Local composition
- Page 91 and 92:
Therefore, the simplified Pitzer eq
- Page 93 and 94:
Parameter Name/Element Symbol Defau
- Page 95 and 96:
Where: �i 2 Thermodynamic Propert
- Page 97 and 98:
The electrolyte NRTL model uses the
- Page 99 and 100:
Parameter Symbol No. of Name Elemen
- Page 101 and 102:
with Where: xi = Mole fraction of c
- Page 103 and 104:
It should be understood that equati
- Page 105 and 106:
The reference Gibbs energy is deter
- Page 107 and 108:
Where: j and k can be any species (
- Page 109 and 110:
The pure component dielectric const
- Page 111 and 112:
2 Thermodynamic Property Models 109
- Page 113 and 114:
�I *PDH = Pitzer-Debye-Hückel te
- Page 115 and 116:
Where: �i Vi = Activity coefficie
- Page 117 and 118:
Recommended cij Values for Differen
- Page 119 and 120:
Parameter Name/ Element 2 Thermodyn
- Page 121 and 122:
NRTL-SAC Reference States The NRTL-
- Page 123 and 124:
2 Thermodynamic Property Models 121
- Page 125 and 126:
For an anionic component, we have M
- Page 127 and 128:
For a molecular segment, the activi
- Page 129 and 130:
Specifically, The long range intera
- Page 131 and 132:
where and are activity coefficients
- Page 133 and 134:
and Y+, are used to reflect the wid
- Page 135 and 136:
Parameter Name/ Element Symbol Defa
- Page 137 and 138:
Reference C.-C. Chen and Y. Song, "
- Page 139 and 140:
Parameter Name GMPTPS, GMPTP1, GMPT
- Page 141 and 142:
zi = Charge of ion i Subscripts c,
- Page 143 and 144:
For option code = 0 (default), the
- Page 145 and 146:
and for n-m electrolytes: 2 Thermod
- Page 147 and 148:
= 0.00979 Perform the necessary con
- Page 149 and 150:
Pitzer, K.S., J.R. Peterson, and L.
- Page 151 and 152:
A4,ij = gij / T + hij A5,ij = mij /
- Page 153 and 154:
Symmetric and Unsymmetric Electroly
- Page 155 and 156:
Parameter Symbol No. of Name Elemen
- Page 157 and 158:
Reference state for ionic component
- Page 159 and 160:
with 2 Thermodynamic Property Model
- Page 161 and 162:
G for these pairs is calculated the
- Page 163 and 164:
with 2 Thermodynamic Property Model
- Page 165 and 166:
2 Thermodynamic Property Models 163
- Page 167 and 168:
Activity Coefficient Basis for Henr
- Page 169 and 170:
Where is the liquid Gibbs free ener
- Page 171 and 172:
Ionic components 2 Thermodynamic Pr
- Page 173 and 174:
the pure fused salts. Applying Eq.
- Page 175 and 176:
�k is the activity coefficient of
- Page 177 and 178:
References U. Weidlich and J. Gmehl
- Page 179 and 180:
ti' = �ij 2 Thermodynamic Propert
- Page 181 and 182:
Wagner Interaction Parameter The Wa
- Page 183 and 184:
References G.M. Wilson, J. Am. Chem
- Page 185 and 186:
Extended Antoine Equation Parameter
- Page 187 and 188:
IK-CAPE Vapor Pressure Equation The
- Page 189 and 190:
Harlacher and Braun, "A Four-Parame
- Page 191 and 192:
Parameter Name/Element 2 Thermodyna
- Page 193 and 194:
General Pure Component Heat of Vapo
- Page 195 and 196:
IK-CAPE Heat of Vaporization Equati
- Page 197 and 198:
Where: xp = Mole fraction of pseudo
- Page 199 and 200:
Parameter Name/Element VLBROC/1 V i
- Page 201 and 202:
Where: Vm l,t = Liquid volume per n
- Page 203 and 204:
Debye-Hückel Volume The Debye-Hüc
- Page 205 and 206:
DIPPR DIPPR equation 105 is the def
- Page 207 and 208:
Rackett The Rackett equation is: Wh
- Page 209 and 210:
Rackett/Campbell-Thodos Mixture Liq
- Page 211 and 212:
Zm RA Zi *,RA Vcm 2 Thermodynamic P
- Page 213 and 214:
General Pure Component Solid Molar
- Page 215 and 216:
Liquid Volume Quadratic Mixing Rule
- Page 217 and 218:
If THRSWT/6 is This equation is use
- Page 219 and 220:
Parameter Symbol Default MDS Lower
- Page 221 and 222:
General Pure Component Ideal Gas He
- Page 223 and 224:
(Other DIPPR equations may sometime
- Page 225 and 226:
General Pure Component Solid Heat C
- Page 227 and 228:
Parameter Name/Element 2 Thermodyna
- Page 229 and 230:
The expression for the liquid mole
- Page 231 and 232:
� Entropy: � Heat capacity: �
- Page 233 and 234:
Parameter Name † /Element CPIXPn/
- Page 235 and 236:
Option Codes for Electrolyte NRTL E
- Page 237 and 238:
Parameter Name Applicable Component
- Page 239 and 240:
For subcritical components: Hm l -H
- Page 241 and 242:
The vapor enthalpy is calculated fr
- Page 243 and 244:
Where: 2 Thermodynamic Property Mod
- Page 245 and 246:
Correlation algorithms for ionic sp
- Page 247 and 248:
Model Model name Phase(s) Pure Mixt
- Page 249 and 250:
Model Type Aspen Liquid Mixture Vis
- Page 251 and 252:
(Other DIPPR equations may sometime
- Page 253 and 254:
Parameter Symbol Default MDS Lower
- Page 255 and 256:
�i = Viscosity of component i (N-
- Page 257 and 258:
Parameter Name/Element 3 Transport
- Page 259 and 260:
Where: The Stockmayer or Lennard-Jo
- Page 261 and 262:
Where: Vcij = �ij = 0 (in almost
- Page 263 and 264:
Where: Vcij = �ij = 0 (in almost
- Page 265 and 266:
Jones-Dole Electrolyte Correction T
- Page 267 and 268:
Letsou-Stiel The Letsou-Stiel model
- Page 269 and 270:
Where: The parameter is the mole fr
- Page 271 and 272:
Reference C.H. Twu, "Internally Con
- Page 273 and 274:
Vcij 3 Transport Property Models 27
- Page 275 and 276:
You must provide parameters for the
- Page 277 and 278:
Linear extrapolation of � *,l ver
- Page 279 and 280:
If TRNSWT/4 is This equation is use
- Page 281 and 282:
Parameter Name/Element 3 Transport
- Page 283 and 284:
Parameter Name/Element 3 Transport
- Page 285 and 286:
Parameter Symbol Default MDS Lower
- Page 287 and 288:
Parameter Name/Element 3 Transport
- Page 289 and 290:
References R.C. Reid, J.M. Prausnit
- Page 291 and 292:
The mixture surface tension is then
- Page 293 and 294:
Parameter Name/Element 3 Transport
- Page 295 and 296:
Where: �solv = Dielectric constan
- Page 297 and 298:
4 Nonconventional Solid Property Mo
- Page 299 and 300:
wij = Mass fraction of the jth cons
- Page 301 and 302:
The oxygen and organic sulfur conte
- Page 303 and 304:
Parameter Name/Element Symbol Defau
- Page 305 and 306:
The complete oxidation of carbon is
- Page 307 and 308:
Parameter Name/Element Symbol Defau
- Page 309 and 310:
The equation for �i dm is good fo
- Page 311 and 312:
5 Property Model Option Codes The f
- Page 313 and 314:
Model Name Option Code 5 Property M
- Page 315 and 316:
Model Name Option Code 5 Property M
- Page 317 and 318:
Soave-Redlich-Kwong Option Codes Th
- Page 319 and 320:
Model Name Option Code HLRELNRT and
- Page 321 and 322:
Model Name Option Code GLRELNRT, GL
- Page 323 and 324:
Index A activity coefficient models
- Page 325 and 326:
ideal gas/DIPPR heat capacity model
- Page 327 and 328:
S Sato-Riedel/DIPPR thermal conduct