Influence of the natural aluminium oxide layer on ... - ALU-WEB.DE
Influence of the natural aluminium oxide layer on ... - ALU-WEB.DE
Influence of the natural aluminium oxide layer on ... - ALU-WEB.DE
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
MEASURING & CONTROL<br />
M<strong>on</strong>itoring <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> extrusi<strong>on</strong> dies<br />
cleaning process by an optic sensor<br />
A. Pascual Formoso 1 ; E. Piñeiro Ben 1 ; L. Herrero Castilla 1 ; C. Domínguez 2 , A. Llobera 2<br />
1 2 Aimen Technology Center, P<strong>on</strong>tevedra, Centro Naci<strong>on</strong>al de Microelectrónica (IMB-CNM, CSIC),<br />
Barcel<strong>on</strong>a, Universidad Autónoma de Barcel<strong>on</strong>a, Bellaterra (Barcel<strong>on</strong>a)<br />
The aim <str<strong>on</strong>g>of</str<strong>on</strong>g> this work has been <str<strong>on</strong>g>the</str<strong>on</strong>g> assessment<br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> use <str<strong>on</strong>g>of</str<strong>on</strong>g> a multiple internal<br />
reflecti<strong>on</strong> sensor in order to m<strong>on</strong>itor <str<strong>on</strong>g>the</str<strong>on</strong>g><br />
advance <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> extrusi<strong>on</strong> dies<br />
cleaning process. The main goal <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g><br />
sensor is that it is able to measure different<br />
initial c<strong>on</strong>centrati<strong>on</strong>s <str<strong>on</strong>g>of</str<strong>on</strong>g> NaOH and it<br />
shows <str<strong>on</strong>g>the</str<strong>on</strong>g> point in which <str<strong>on</strong>g>the</str<strong>on</strong>g> aluminate<br />
starts to precipitate allowing to distinguish<br />
states <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> bath <strong>on</strong> c<strong>on</strong>centrati<strong>on</strong><br />
ratios <str<strong>on</strong>g>of</str<strong>on</strong>g> [Al] / [NaOH] = 0.1.<br />
The <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> extrusi<strong>on</strong> industry has a significant<br />
ec<strong>on</strong>omic importance all over Europe.<br />
The process is carried out using extrusi<strong>on</strong> dies,<br />
steel disks with an opening cut through <str<strong>on</strong>g>the</str<strong>on</strong>g>m,<br />
with <str<strong>on</strong>g>the</str<strong>on</strong>g> size and shape <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> intended crosssecti<strong>on</strong><br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> final extruded product. An <str<strong>on</strong>g>aluminium</str<strong>on</strong>g><br />
billet pre-heated (500ºC) is forced<br />
to flow through <str<strong>on</strong>g>the</str<strong>on</strong>g> die in order to get <str<strong>on</strong>g>the</str<strong>on</strong>g><br />
required pr<str<strong>on</strong>g>of</str<strong>on</strong>g>ile. At <str<strong>on</strong>g>the</str<strong>on</strong>g> end <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> cycle, it is<br />
necessary to remove all <str<strong>on</strong>g>the</str<strong>on</strong>g> remaining metal<br />
inside <str<strong>on</strong>g>the</str<strong>on</strong>g> cavities <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> die to reuse it in new<br />
pieces producti<strong>on</strong>. In fact, <str<strong>on</strong>g>the</str<strong>on</strong>g> extrusi<strong>on</strong> dies<br />
are resp<strong>on</strong>sible for quality and process performance<br />
and <str<strong>on</strong>g>the</str<strong>on</strong>g>ir cost is between 35-50% <str<strong>on</strong>g>of</str<strong>on</strong>g><br />
<str<strong>on</strong>g>the</str<strong>on</strong>g> cost <str<strong>on</strong>g>of</str<strong>on</strong>g> manufacture (Guía Tecnológica).<br />
The ordinary cleaning method c<strong>on</strong>sists <str<strong>on</strong>g>of</str<strong>on</strong>g><br />
loading <str<strong>on</strong>g>the</str<strong>on</strong>g> dies into open tanks, c<strong>on</strong>taining a<br />
soluti<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> NaOH (caustic soda) at high c<strong>on</strong>centrati<strong>on</strong><br />
and temperature near <str<strong>on</strong>g>the</str<strong>on</strong>g> boiling<br />
point. The cleaning process may even exceed<br />
10 hours, depending <strong>on</strong> die size and <str<strong>on</strong>g>aluminium</str<strong>on</strong>g><br />
quantity adhered to its surface. Generally,<br />
<str<strong>on</strong>g>the</str<strong>on</strong>g> baths are changed after each use without<br />
making a preliminary analysis to determine<br />
if <str<strong>on</strong>g>the</str<strong>on</strong>g> bath still retains its cleaning properties.<br />
Daily bath emptying (which can exceed 3,000<br />
litre per day) causes a high cost due to <str<strong>on</strong>g>the</str<strong>on</strong>g><br />
new formulati<strong>on</strong> reagents (water and sodium<br />
hydr<str<strong>on</strong>g>oxide</str<strong>on</strong>g>) and energy. In additi<strong>on</strong>, it generates<br />
a large amount <str<strong>on</strong>g>of</str<strong>on</strong>g> hazardous waste (with<br />
high basicity and high c<strong>on</strong>tent <str<strong>on</strong>g>of</str<strong>on</strong>g> dissolved<br />
<str<strong>on</strong>g>aluminium</str<strong>on</strong>g>) (Guía Tecnológica).<br />
Several studies (Li et al, 2005a) have shown<br />
that <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> in a caustic soluti<strong>on</strong> at a temperature<br />
approaching to boiling point leads<br />
to sodium aluminate (NaAlO 2), resulting in<br />
hydrogen gas (H 2 (g)). Sodium aluminate so-<br />
luti<strong>on</strong>s begin to run out, since a mesoscopic<br />
point <str<strong>on</strong>g>of</str<strong>on</strong>g> view, due to presence <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>aluminium</str<strong>on</strong>g><br />
hydr<str<strong>on</strong>g>oxide</str<strong>on</strong>g>s like gibbsite and bayerite (Harris<br />
et al, 1999). Optical scattering techniques, like<br />
Dynamic Light Scattering (DLS), have been<br />
proven its feasibility to characterize <str<strong>on</strong>g>the</str<strong>on</strong>g> nucleati<strong>on</strong><br />
and growth <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g>se species.<br />
The growing interest <str<strong>on</strong>g>of</str<strong>on</strong>g> industry for c<strong>on</strong>trol<br />
and m<strong>on</strong>itoring <str<strong>on</strong>g>of</str<strong>on</strong>g> producti<strong>on</strong> process<br />
motivates research in <str<strong>on</strong>g>the</str<strong>on</strong>g> development <str<strong>on</strong>g>of</str<strong>on</strong>g> new<br />
optical devices that allow <str<strong>on</strong>g>the</str<strong>on</strong>g>ir integrati<strong>on</strong> into<br />
producti<strong>on</strong> lines (Harris et al, 1999). These<br />
systems provide real-time informati<strong>on</strong> about<br />
a process. One <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g>se labs <strong>on</strong> a chip device<br />
are <str<strong>on</strong>g>the</str<strong>on</strong>g> multiple internal reflecti<strong>on</strong> (MIR) systems.<br />
(Llobera et al, 2007).<br />
The aim <str<strong>on</strong>g>of</str<strong>on</strong>g> this work has been <str<strong>on</strong>g>the</str<strong>on</strong>g> assessment<br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> use <str<strong>on</strong>g>of</str<strong>on</strong>g> a low cost MIR sensor<br />
fabricated in PDMS in order to m<strong>on</strong>itor<br />
<str<strong>on</strong>g>the</str<strong>on</strong>g> advance <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> extrusi<strong>on</strong> dies<br />
cleaning process.<br />
Method<br />
Soluti<strong>on</strong> preparati<strong>on</strong>: Due to <str<strong>on</strong>g>the</str<strong>on</strong>g> impossibility<br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> working with real extrusi<strong>on</strong> dies at laboratory<br />
scale, small pieces <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> have<br />
been used for <str<strong>on</strong>g>the</str<strong>on</strong>g> simulati<strong>on</strong>s. Cleaning baths<br />
are prepared using NaOH soluti<strong>on</strong> in a benchscale.<br />
This bath is heated to <str<strong>on</strong>g>the</str<strong>on</strong>g> temperature<br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> work and <str<strong>on</strong>g>the</str<strong>on</strong>g> fragment <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> is put<br />
into <str<strong>on</strong>g>the</str<strong>on</strong>g> bath. Once treatment time is completed,<br />
<str<strong>on</strong>g>the</str<strong>on</strong>g> attacked <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> piece is removed.<br />
When <str<strong>on</strong>g>the</str<strong>on</strong>g> bath is cool<br />
a dark gelatinous compounds<br />
start to precipitate<br />
(<str<strong>on</strong>g>aluminium</str<strong>on</strong>g><br />
hydr<str<strong>on</strong>g>oxide</str<strong>on</strong>g> and aluminates).<br />
The <str<strong>on</strong>g>aluminium</str<strong>on</strong>g><br />
c<strong>on</strong>centrati<strong>on</strong> in soluti<strong>on</strong><br />
and free sodium<br />
hydr<str<strong>on</strong>g>oxide</str<strong>on</strong>g> c<strong>on</strong>centrati<strong>on</strong><br />
are determined in<br />
each test <str<strong>on</strong>g>of</str<strong>on</strong>g> experimental<br />
set-up. The ratio <str<strong>on</strong>g>of</str<strong>on</strong>g><br />
free sodium hydr<str<strong>on</strong>g>oxide</str<strong>on</strong>g><br />
and <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> in so-<br />
luti<strong>on</strong> gives an idea <str<strong>on</strong>g>of</str<strong>on</strong>g><br />
bath exhausti<strong>on</strong>.<br />
Optical characterizati<strong>on</strong>: This work has been<br />
carried out to characterize <str<strong>on</strong>g>the</str<strong>on</strong>g> optical properties<br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> samples such as refractive index, transmittance<br />
spectral resp<strong>on</strong>se and scattering.<br />
The size distributi<strong>on</strong> has been analyzed by<br />
dynamic light scattering, from samples prepared<br />
according to <str<strong>on</strong>g>the</str<strong>on</strong>g> procedure menti<strong>on</strong>ed<br />
previously (to 95ºC from a soluti<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> initial<br />
NaOH c<strong>on</strong>centrati<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> 19%). The size <str<strong>on</strong>g>of</str<strong>on</strong>g> particles<br />
suspended in <str<strong>on</strong>g>the</str<strong>on</strong>g> bath has been determined<br />
using a Spectrometer Autosizer 4800<br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> Malvern Instruments (PCS technique). The<br />
refractive index has been measured at different<br />
times (with a refractometer ABBEMAT-<br />
HP) for so what‘s <str<strong>on</strong>g>the</str<strong>on</strong>g> bath depleti<strong>on</strong> point.<br />
Reference measurement system: Experiments<br />
with laboratory samples have been<br />
c<strong>on</strong>ducted to evaluate <str<strong>on</strong>g>the</str<strong>on</strong>g> behaviour <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g><br />
samples measured by <str<strong>on</strong>g>the</str<strong>on</strong>g> sensor and verify<br />
<str<strong>on</strong>g>the</str<strong>on</strong>g> feasibility <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> proposed detecti<strong>on</strong><br />
methods, as well as <str<strong>on</strong>g>the</str<strong>on</strong>g> needs and tolerances<br />
<str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>the</str<strong>on</strong>g> coupling and sealing systems. For <str<strong>on</strong>g>the</str<strong>on</strong>g>se<br />
tests, a setup similar to <str<strong>on</strong>g>the</str<strong>on</strong>g> proposed sensor<br />
has been used, but discrete optical elements<br />
were used instead <str<strong>on</strong>g>of</str<strong>on</strong>g> waveguides, as a reference<br />
measurement system. This assembly allows<br />
to develop transmissi<strong>on</strong> and dispersi<strong>on</strong><br />
measurements at 90° simultaneously. The light<br />
is injected through a multimode optical fibre<br />
and collected with plastic optical fibres <str<strong>on</strong>g>of</str<strong>on</strong>g> 1<br />
mm in diameter.<br />
In order to improve <str<strong>on</strong>g>the</str<strong>on</strong>g> signal to noise ratio<br />
and eliminate <str<strong>on</strong>g>the</str<strong>on</strong>g> c<strong>on</strong>tributi<strong>on</strong> <str<strong>on</strong>g>of</str<strong>on</strong>g> light which is<br />
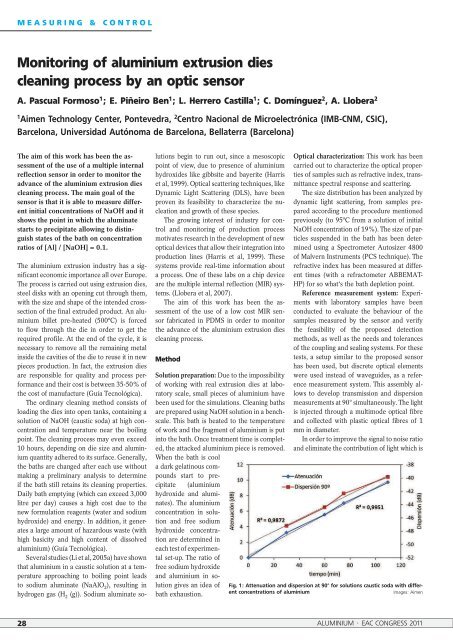
Fig. 1: Attenuati<strong>on</strong> and dispersi<strong>on</strong> at 90° for soluti<strong>on</strong>s caustic soda with different<br />
c<strong>on</strong>centrati<strong>on</strong>s <str<strong>on</strong>g>of</str<strong>on</strong>g> <str<strong>on</strong>g>aluminium</str<strong>on</strong>g> Images: Aimen<br />
28 <strong>ALU</strong>MINIUM · EAC CONGRESS 2011