Drosophila - Severo Ochoa - Universidad Autónoma de Madrid
Drosophila - Severo Ochoa - Universidad Autónoma de Madrid
Drosophila - Severo Ochoa - Universidad Autónoma de Madrid
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Jefe <strong>de</strong> Línea /<br />
Group Lea<strong>de</strong>r:<br />
Jesús Ávila <strong>de</strong> Grado<br />
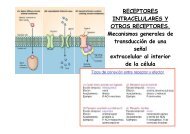
Función <strong>de</strong> las proteínas<br />
microtubulares en neuronas<br />
Function of microtubular proteins<br />
in neurons<br />
D2<br />
Resumen <strong>de</strong> investigación<br />
Research summary<br />
CBM 2005/2006<br />
Personal Científico /<br />
Scientific Staff:<br />
María Teresa Moreno-Flores<br />
Félix Hernán<strong>de</strong>z<br />
Filip Lim<br />
Mar Pérez<br />
90<br />
Postdoctorales /<br />
Postdoctoral:<br />
Alberto Gómez<br />
Eva-María Jiménez<br />
Tobías Engel<br />
Becarios Predoctorales /<br />
Predoctoral Fellows:<br />
Alicia Rubio<br />
Paloma Goñi<br />
Elena Tortosa<br />
Elena Gómez <strong>de</strong> Barreda<br />
Técnicos <strong>de</strong> Investigación /<br />
Technical Assistance:<br />
Raquel Cuadros<br />
Elena Langa<br />
Santiago Soto-Largo<br />
María Jesús Bermejo<br />
Esther García<br />
Marta Ramos<br />
Neurobiología Neurobiology<br />
Nuestro trabajo ha continuado con el análisis funcional <strong>de</strong><br />
las proteínas a) MAP1B y b) tau, y las consecuencias <strong>de</strong> su<br />
falta <strong>de</strong> función o función aberrante en procesos<br />
neuro<strong>de</strong>generativos. Adicionalmente hemos seguido con el<br />
estudio c) <strong>de</strong> la reparación axonal en algunos sistemas<br />
mo<strong>de</strong>lo.<br />
a) Nuestros resultados sobre MAP1B han indicado que una<br />
función <strong>de</strong> esta proteína es la <strong>de</strong> modular los procesos <strong>de</strong><br />
transporte axonal retrógrado <strong>de</strong> las mitocondrias.<br />
Adicionalmente, hemos observado que la carencia <strong>de</strong><br />
MAP1B pue<strong>de</strong> ser complementada, en parte, por otra<br />
proteína que se asocia a los microtúbulos, EB-1.<br />
b) Sobre la proteína tau nuestro trabajo ha continuado el<br />
estudio in vitro <strong>de</strong> su fosforilación y <strong>de</strong> su polimerización.<br />
La posible relación entre fosforilación y agregación <strong>de</strong> tau<br />
se ha analizado in vitro, en cultivos celulares y en mo<strong>de</strong>los<br />
<strong>de</strong> <strong>Drosophila</strong> y <strong>de</strong> ratón. En estos últimos, utilizando<br />
animales doble transgénicos condicionales que<br />
sobreexpresan la proteína quinasa que mayoritariamente<br />
modifica a tau, y la proteína tau con alguna <strong>de</strong> las<br />
mutaciones que aparecen en algunas tauopatías, hemos<br />
observado que algunas <strong>de</strong> las patologías inducidas en el<br />
ratón, como son la hiperfosforilación y la agregación <strong>de</strong> tau,<br />
son reversibles (fosforilación), mientras que otras<br />
(formación <strong>de</strong> agregados aberrantes), no lo son.<br />
c) Con respecto a nuestros estudios sobre el uso <strong>de</strong> células<br />
<strong>de</strong> glía envolvente, como reparadores <strong>de</strong>l daño axonal,<br />
hemos seguido caracterizando dichas células <strong>de</strong> glía<br />
envolvente, fundamentalmente <strong>de</strong>s<strong>de</strong> un punto <strong>de</strong> vista<br />
funcional. Hemos inmortalizado estas células y hemos<br />
estudiado su funcionalidad.<br />
Adicionalmente, hemos comparado la expresión diferencial<br />
<strong>de</strong> genes en células <strong>de</strong> glía con gran actividad reparadora<br />
con otras que carecen <strong>de</strong> esta actividad. Entre los<br />
productos que pudieran estar implicados en el proceso <strong>de</strong><br />
regeneración hemos encontrado a la proteína MMP2 y al<br />
factor <strong>de</strong> crecimiento BNDF.<br />
Figura 1. Presencia <strong>de</strong> la proteína tau hiperfosforilada en ratones doble<br />
transgénicos que sobrexpresan la quinasa GSK-3β y la proteína tau con tres<br />
mutaciones presentes en enfermos <strong>de</strong> FTDP-17 (ratones GSK-3/VLW), tratados con<br />
bajas dosis <strong>de</strong> LiCl. Pue<strong>de</strong> observarse tau hiperfosforilado en la región CA1 <strong>de</strong> los<br />
animales GSK-3/VLW controles; sin embargo, en animales tratados con litio es<br />
in<strong>de</strong>tectable la presencia <strong>de</strong> la proteína tau hiperfosforilada. Barra = 100 µm.<br />
Figure 1. Phosphorylation state of tau in mice overexpressing the kinase GSK-3β<br />
and tau protein with three FTDP-17 missense mutations (GSK-3/VLW mice) treated<br />
with a low dose of LiCl. High levels of hyperphosphorylated somato<strong>de</strong>n<strong>de</strong>ndritic tau<br />
in CA1 of GSK-3/VLW control mice was evi<strong>de</strong>nt, while in mice treated with lithium,<br />
immuno<strong>de</strong>tection of tau phosphorylation was absent. Scale bar = 100 µm.<br />
We have continued with the functional analyses of MAP1B<br />
and tau proteins, and the consequences of their lack or<br />
aberrant function in neuro<strong>de</strong>generative disor<strong>de</strong>rs.<br />
Additionally, we have continued with the study of the axonal<br />
repair in some mo<strong>de</strong>ls.<br />
Our results about MAP1B have indicated that the function of<br />
this protein is to modulate retrogra<strong>de</strong> axonal transport<br />
processes in mitochondria, being this transport <strong>de</strong>creased<br />
with the presence of this protein.<br />
Additionally, we have observed that the lack of MAP1B could<br />
be complemented by another protein that also is associated<br />
to microtubules, EB-1.<br />
Concerning tau protein, we have followed the in vitro studies<br />
about its polymerization and phosphorylation.<br />
The possible relation between phosphorylation and<br />
aggregation has been analyzed in vitro, in cellular cultures, in<br />
<strong>Drosophila</strong> and in mice. We have worked with double<br />
transgenic conditional mice which overexpress the kinase<br />
protein that majority modifies tau, and also express tau<br />
protein with some of its mutations present in several<br />
tauopathies. We have observed that some induced<br />
pathologies in mice, like tau hyperphosphorylation and<br />
aggregation, are in some cases reversible (phosphorylation)<br />
whereas in others (formation of aberrant aggregates) are not.<br />
About our studies on ensheathing olfactory glia cells as<br />
reparators of axonal damage, we have continued with the<br />
characterization of these cells, especially from the functional<br />
point of view. We have immortalized these cells and studied<br />
their functionality after immortalization.<br />
Additionally, we have compared the differential expression of<br />
the genes from glia cells with a high restoring activity with those<br />
from cells lacking such activity. We have i<strong>de</strong>ntified that at least<br />
two proteins could be implicated in the regeneration process;<br />
these are MMP2 protein and the growing factor BNDF.<br />
Publicaciones seleccionadas<br />
Selected publications<br />
Ávila, J. (2006). Tau phosphorylation and aggregation in Alzheimer's disease pathology. FEBS Lett. 580, 2922-2927.<br />
Ávila, J. (2006). Tau protein, the main component of paired helical filaments. J Alzheimers Dis. 9, 171-175.<br />
Ávila, J., Nitsch, R.M., Haass, C. and De Strooper, B. (2006) European Alzheimer disease funding. Nat. Med. 12,<br />
776-777.<br />
Engel, T., Goni-Oliver, P., Lucas, J.J., Ávila, J. and Hernán<strong>de</strong>z, F. (2006). Chronic lithium administration to FTDP-17<br />
tau and GSK-3beta overexpressing mice prevents tau hyperphosphorylation and neurofibrillary tangle formation,<br />
but pre-formed neurofibrillary tangles do not revert. J Neurochem. 99, 1445-1455.<br />
Engel, T., Hernán<strong>de</strong>z, F., Ávila, J. and Lucas, J.J. (2006). Full reversal of Alzheimer's disease-like phenotype in a<br />
mouse mo<strong>de</strong>l with conditional overexpression of glycogen synthase kinase-3. J. Neurosci. 26, 5083-5090.<br />
Engel, T., Lucas, J.J., Gomez-Ramos, P., Moran, M.A., Ávila, J. and Hernán<strong>de</strong>z, F. (2006). Cooexpression of FTDP-<br />
17 tau and GSK-3beta in transgenic mice induce tau polymerization and neuro<strong>de</strong>generation. Neurobiol. Aging 27,<br />
1258-1268.<br />
Farias, G.G., Godoy, J.A., Vázquez, M.C., Adani, R., Meshulam, H., Ávila, J., Amitai, G, and Inestrosa, N.C. (2005).<br />
The anti-inflammatory and cholinesterase inhibitor bifunctional compound IBU-PO protects from beta-amyloid<br />
neurotoxicity by acting on Wnt signaling components. Neurobiol Dis, 18, 176-183.<br />
Ferrer, I., Barrachina, M., Puig, B., Martínez <strong>de</strong> Lagran, M., Marti, E., Ávila, J. and Dierssen, M. (2005). Constitutive<br />
Dyrk1A is abnormally expressed in Alzheimer disease, Down syndrome, Pick disease, and related transgenic<br />
mo<strong>de</strong>ls. Neurobiol Dis. 20, 392-400.<br />
Gasic, G.P., Barco, A., Ávila, J. and Lerma, J. (2006). A meeting to remember: meeting on memory and related<br />
disor<strong>de</strong>rs. EMBO Rep. 7, 768-773.<br />
Gomez-Ramos, A., Díaz-Hernán<strong>de</strong>z, M., Cuadros, R., Hernán<strong>de</strong>z, F. and Ávila, J. (2006). Extracellular tau is toxic to<br />
neuronal cells. FEBS Lett. 580, 4842-4850.<br />
Gomez-Ramos, A., Dominguez, J., Zafra, D., Corominola, H., Gomis, R., Guinovart, J.J. and Ávila, J. (2006).<br />
Sodium tungstate <strong>de</strong>creases the phosphorylation of tau through GSK3 inactivation. J. Neurosci Res. 83, 264-273.<br />
González-Billault, C., Del Río, J.A., Urena, J.M., Jiménez-Mateos, E.M., Barallobre, M.J., Pascual, M., Pujadas, L.,<br />
Simo, S., Torre, A.L., Gavin, R., WandosJiménezell, F., Soriano, E, and Ávila, J. (2005). A role of MAP1B in Reelin<strong>de</strong>pen<strong>de</strong>nt<br />
neuronal migration. Cereb. Cortex 15, 1134-1145.<br />
Jiménez-Mateos, E.M., González-Billault, C., Dawson, H.N., Vitek, M.P. and<br />
Ávila, J. (2006). Role of MAP1B in axonal retrogra<strong>de</strong> transport of<br />
mitochondria. Biochem. J. 397, 53-59.<br />
Jiménez-Mateos, E.M., Paglini, G., González-Billault, C., Caceres, A. and<br />
Ávila J (2005). End binding protein-1 (EB1) complements microtubuleassociated<br />
protein-1B during axonogenesis. J. Neurosci. Res. 80, 350-359.<br />
Jiménez-Mateos, E.M., Wandosell, F., Reiner, O., Ávila, J. and González-<br />
Billault, C. (2005). Binding of microtubule-associated protein 1B to LIS1<br />
affects the interaction between dynein and LIS1. Biochem. J. 389, 333-341.<br />
Kunjithapatham, R., Oliva, F.Y., Doshi, U., Pérez, M., Ávila, J. and Munoz, V.<br />
(2005). Role for the alpha-helix in aberrant protein aggregation.<br />
Biochemistry 44, 149-156.<br />
Liu, Q., Lee, H.G., Honda, K., Siedlak, S.L., Harris, P.L., Cash, A.D., Zhu, X.,<br />
Ávila, J., Nunomura, A., Takeda, A,. Smith, M.A. and Perry, G. (2005). Tau<br />
modifiers as therapeutic targets for Alzheimer's disease. Biochim Biophys<br />
Acta, 1739, 211-215.<br />
Mendieta, J., Fuertes, M.A., Kunjishapatham, R., Santa-María, I., Moreno,<br />
F.J., Alonso, C., Gago, F., Munoz, V., Ávila, J.and Hernán<strong>de</strong>z, F. (2005).<br />
Phosphorylation modulates the alpha-helical structure and polymerization of<br />
a pepti<strong>de</strong> from the third tau microtubule-binding repeat. Biochim Biophys<br />
Acta 1721, 16-26.<br />
Moreno-Flores, M.T., Bradbury, E.J., Martín-Bermejo, M.J., Agudo, M., Lim, F.,<br />
Pastrana, E., Ávila, J., Díaz-Nido, J., McMahon, S.B. and Wandosell, F. (2006).<br />
A clonal cell line from immortalized olfactory ensheathing glia promotes<br />
functional recovery in the injured spinal cord. Mol. Ther. 13, 598-608.<br />
Pastrana, E., Moreno-Flores, M.T., Gurzov, E.N., Ávila, J., Wandosell, F. and<br />
Díaz Nido, J. (2006). Genes associated with adult axon regeneration<br />
promoted by olfactory ensheathing cells: a new role for matrix<br />
metalloproteinase 2. J. Neurosci. 26, 5347-5359.<br />
Pérez, M., Ribe, E., Rubio, A., Lim, F., Moran, M.A,. Ramos, P.G., Ferrer, I.,<br />
Isla, M.T. and Ávila, J. (2005). Characterization of a double (amyloid<br />
precursor protein-tau) transgenic: tau phosphorylation and aggregation.<br />
Neuroscience 130, 339-347.<br />
Ribe, E.M., Pérez, M., Puig, B., Gich, I., Lim, F., Cuadrado, M., Sesma, T.,<br />
Catena, S., Sanchez, B., Nieto, M., Gomez-Ramos, P., Moran, M.A.,<br />
Cabo<strong>de</strong>villa, F., Samaranch, L., Ortiz, L., Pérez, A., Ferrer, I., Ávila, J. and<br />
Gomez-Isla, T. (2005). Accelerated amyloid <strong>de</strong>position, neurofibrillary<br />
<strong>de</strong>generation and neuronal loss in double mutant APP/tau transgenic mice.<br />
Neurobiol Dis. 20, 814-822.<br />
Ros, R., Thobois, S., Streichenberger, N., Kopp, N., Sanchez, M.P., Pérez,<br />
M., Hoenicka, J., Ávila, J., Honnorat, J. and <strong>de</strong> Yebenes, J.G. (2005). A new<br />
mutation of the tau gene, G303V, in early-onset familial progressive<br />
supranuclear palsy. Arch Neurol. 62, 1444-1450..<br />
Santa-María, I., Pérez, M., Hernán<strong>de</strong>z, F., Ávila, J. and Moreno, F.J. (2006).<br />
Characteristics of the binding of thioflavin S to tau paired helical filaments. J.<br />
Alzheimers Dis. 9, 279-285.<br />
Santa-María, I., Pérez, M., Hernán<strong>de</strong>z, F., Munoz, V., Moreno, F.J. and Ávila,<br />
J. (2006). In vitro tau fibrillization: mapping protein regions. Biochim Biophys<br />
Acta 1762, 683-692.<br />
Santa-María, I., Smith, M.A., Perry, G., Hernán<strong>de</strong>z, F., Ávila, J. and Moreno,<br />
F.J. (2005). Effect of quinones on microtubule polymerization: a link between<br />
oxidative stress and cytoskeletal alterations in Alzheimer's disease. Biochim<br />
Biophys Acta 1740, 472-480.<br />
Smith, M.A., Nunomura, A., Lee, H.G., Zhu, X., Moreira, P.I., Ávila, J. and<br />
Perry, G. (2005). Chronological primacy of oxidative stress in Alzheimer<br />
disease. Neurobiol Aging 26, 579-580.<br />
Tesis doctorales<br />
Doctoral Theses<br />
Eva María Jiménez. (2005) Estudio funcional <strong>de</strong> la proteína asociada a<br />
microtúbulos MAP1B”. <strong>Universidad</strong> Autónoma <strong>de</strong> <strong>Madrid</strong>. Sobresaliente cum<br />
lau<strong>de</strong>. Dirigida por Jesús Ávila.<br />
Tobias Engel. (2005) Estudio <strong>de</strong>l posible sinergismo en la<br />
neuro<strong>de</strong>generación inducida por sobreexpresión <strong>de</strong> GSK-3β y por<br />
mutaciones <strong>de</strong> tau mediante coexpresión en mo<strong>de</strong>los <strong>de</strong> ratón. <strong>Universidad</strong><br />
Autónoma <strong>de</strong> <strong>Madrid</strong>. Sobresaliente cum lau<strong>de</strong>. Codirigida por Félix<br />
Hernán<strong>de</strong>z y José J. Lucas.<br />
Premios<br />
Awards<br />
National Prize Caja Rural “Ciudad <strong>de</strong> Zamora”, 2005.<br />
Organización <strong>de</strong> reuniones<br />
Meetings organizations<br />
10th International Conference on Alzheimer’s Disease and Related<br />
Disor<strong>de</strong>rs. (July, 2006).<br />
Participación en los Actos <strong>de</strong> Homenaje a D. Santiago Ramón y Cajal, en el<br />
centenario <strong>de</strong> la Concesión <strong>de</strong>l Premio Nobel.<br />
91